| CATEGORII DOCUMENTE |
| Bulgara | Ceha slovaca | Croata | Engleza | Estona | Finlandeza | Franceza |
| Germana | Italiana | Letona | Lituaniana | Maghiara | Olandeza | Poloneza |
| Sarba | Slovena | Spaniola | Suedeza | Turca | Ucraineana |
Myocardial Infarction
3.1. Definition, Etiology, and Pathophysiology
Myocardial infarction refers to the process by which myocardial tissue is destroyed in regions of the heart that are deprived of an adequate blood supply because of a reduced coronary blood flow. The cause of the reduced blood flow is either a critical narrowing of a coronary artery due to atherosclerosis or a complete occlusion of an artery due to embolus or thrombus. Decreased coronary blood flow may also result
from shock and hemorrhage. In each case, there is a profound imbalance between myocardial oxygen supply and demand.
,,Coronary occlusion, ,, heart attack and ,,myocardial infarction are all used synonymously, but the preferred term is myocardial infarcti
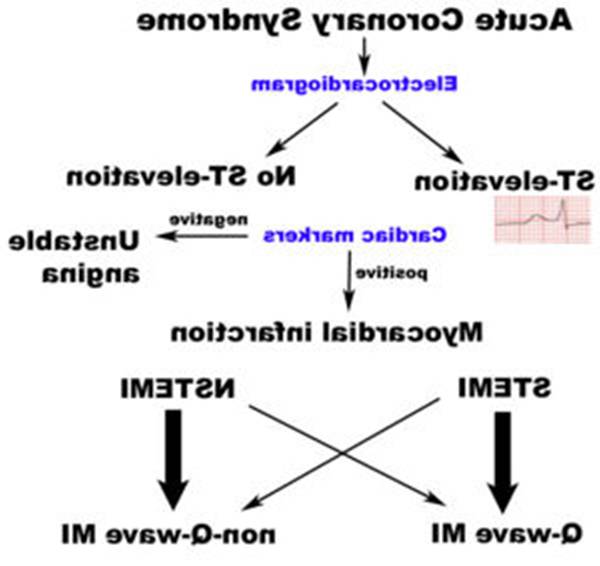
Acute myocardial infarction is a type of acute coronary syndrome, which is most frequently (but not always) a manifestation of coronary artery disease. The acute coronary syndromes include ST segment elevation myocardial infarction (STEMI), non-ST segment elevation myocardial infarction (NSTEMI), and unstable angina (UA).
Depending on the location of the obstruction in the coronary circulation, different zones of the heart can become injured. Using the anatomical terms of location, one can
describe anterior, inferior, lateral, apical and septal infarctions (and combinations, such as anteroinferior, anterolateral, and so on). For example, an occlusion of the left anterior descending coronary artery will result in an anterior wall myocardial infarct.
Another distinction is whether a MI is subendocardial, affecting only the inner third to one half of the heart muscle, or transmural, damaging (almost) the entire wall of the heart. The inner part of the heart muscle is more vulnerable to oxygen shortage, because the coronary arteries run inward from the epicardium to the endocardium, and because the blood flow through the heart muscle is hindered by the heart contraction.
The phrases transmural and subendocardial infarction used to be considered synonymous with Q-wave and non-Q-wave myocardial infarction respectively, based on the presence or absence of Q waves on the ECG. It has since been shown that there is no clear correlation between the presence of Q waves with a transmural infarction and the absence of Q waves with a subendocardial infarction, but Q waves are associated with larger infarctions, while the lack of Q waves is associated with smaller infarctions. The presence or absence of Q-waves also has clinical importance, with improved outcomes associated with a lack of Q waves.
The phrase 'massive attack' is not a recognized medical term.
3.3. Clinical Manifestation
The patient with myocardial infarction is usually male, over 40, and has atherosclerosis of the coronary vessels, often with arterial hypertension. Attacks also occur in women and in younger men in their early 30s or even 20s. Women who take oral contraceptives and also smoke are at very high risk. Overall, however, the rate of myocardial infarction is greater in men than in women at all ages.
Continuous chest pain, characterized by a sudden onset, usually over the lower sternal region and the upper abdomen, is the primary presenting symptom. The pain may increase steadily in severity until it becomes almost unbearable. It is a heavy, viselike pain, which may radiate to the shoulders and down the arms, usually the left arm. Unlike the pain of true angina, it begins spontaneously (not after effort, emotional upset, and the like), persists for hours or days, and is relieved neither by rest nor by nitroglycerin. In some cases the pain may radiate to the jaw and neck. The pain is often accompanied by shortness of breath; pallor; cold, clammy diaphoresis; dizziness or light-headedness; and nausea and vomiting.
The patient with diabetes mellitus may not experience severe pain with myocardial infarction. The neuropathy that accompanies diabetes can interfere with neuroreceptors, thus dulling the pain experience.
3.4. Gerontologic Consideration
The elderly patient may not experience the typical viselike pain associated with myocardial infarction because of the diminished responses of neurotransmitters that occur in the aging process. Often the pain is atypical, such as jaw pain, or fainting may be experienced.
The arteriosclerosis that accompanies aging may compromise tissue perfusion because of increased peripheral vascular resistance. Because elderly patients may have
a well established collateral circulation of the myocardium, they often are spared the lethal complications associated with myocardial infarction.
3.5. Diagnostic Evaluation
Diagnosis of myocardial infarction is generally based on history of the present illness, electrocardigram, and serial serum enzymes. Prognosis depends on the severity of coronary artery obstruction and hence the extent of myocardial damage. Physical examination is always conducted but alone is insufficient to confirm diagnosis.
Patient history. The taking of a patient history occurs in two steps:
the history of the present illness
the history of previous illnesses and family health history, particularly related to the incidence of heart disease in the family
Previous history often can provide valuable information about the patient s risk factors or coronary heart disease. The history of the present illness is in many cases conclusive for the diagnosis of myocardial infarction.
The patient s history provides subjective data. The careful practitioner follows the history with interpretation of objective data offered by the ECG and serial enzyme studies.
The Electrocardiogram. The ECG provides information about the electrophysiology of the heart. Through the use of serial readings, the physician is able to monitor the evolution and resolution of an MI. The location and relative size of the infarction also may be determined by ECG.
The primary purpose of the electrocardiogram is to detect ischemia or acute coronary injury in broad, symptomatic emergency department populations. However, the standard 12 lead ECG has several limitations. An ECG represents a brief sample in time. Because unstable ischemic syndromes have rapidly changing supply versus demand characteristics, a single ECG may not accurately represent the entire picture. It
is therefore desirable to obtain serial 12 lead ECGs, particularly if the first ECG is obtained during a pain-free episode. Alternatively, many emergency departments and chest pain centers use computers capable of continuous ST segment monitoring.It should also be appreciated that the standard 12 lead ECG does not directly examine the right ventricle, and does a relatively poor job of examining the posterior basal and lateral walls of the left ventricle. In particular, acute myocardial infarction in the distribution of the circumflex artery is likely to produce a nondiagnostic ECG.The use of non-standard ECG leads like right-sided lead V4R and posterior leads V7, V8, and V9 may improve sensitivity for right ventricular and posterior myocardial infarction. In
spite of these limitations, the 12 lead ECG stands at the center of risk stratification for the patient with suspected acute myocardial infarction. Mistakes in interpretation are relatively common, and the failure to identify high risk features has a negative effect on the quality of patient care.The 12 lead ECG is used to classify patients into one of three groups:
1. those with ST segment elevation or new bundle branch block (suspicious for acute injury and a possible candidate for acute reperfusion therapy with thrombolytics or primary PCI),
2. those with ST segment depression or T wave inversion (suspicious for ischemia), and
3. those with a so-called non-diagnostic or normal ECG.
A normal ECG does not rule out acute myocardial infarction. Sometimes the earliest presentation of acute myocardial infarction is the hyperacute T wave, which is treated the same as ST segment elevation.In practice this is rarely seen, because it only exists for 2-30 minutes after the onset of infarction. Hyperacute T waves need to be distinguished from the peaked T waves associated with hyperkalemia. The current guidelines for the ECG diagnosis of acute myocardial infarction require at least 1 mm (0.1 mV) of ST segment elevation in 2 or more anatomically contiguous leads. This criterion is problematic, however, as acute myocardial infarction is not the most
common cause of ST segment elevation in chest pain patients.In addition, over 90% of healthy men have at least 1 mm (0.1 mV) of ST segment elevation in at least one precordial lead. The clinician must therefore be well versed in recognizing the so-called ECG mimics of acute myocardial infarction, which include left ventricular hypertrophy, left bundle branch block, paced rhythm, benign early repolarization, pericarditis, hyperkalemia, and ventricular aneurysm.
Serum Enzymes and Isoenzymes Serial enzyme studies include the following
Creatine Kinase - Total: The total CK is a simple and inexpensive test that is readily available using many laboratory instruments. However, an elevation in total CK
is not specific for myocardial injury, because most CK is located in skeletal muscle, and elevations are possible from a variety of non-cardiac conditions.
Creatine Kinase - MB Fraction Creatine kinase can be further subdivided into three isoenzymes: MM, MB, and BB. The MM fraction is present in both cardiac and skeletal muscle, but the MB fraction is much more specific for cardiac muscle: about 15 to 40% of CK in cardiac muscle is MB, while less than 2% in skeletal muscle is MB. The BB fraction (found in brain, bowel, and bladder) is not routinely measured. Thus, CK-MB is a very good marker for acute myocardial injury, because of its excellent specificity, and it rises in serum within 2 to 8 hours of onset of acute myocardial infarction. Serial measurements every 2 to 4 hours for a period of 9 to 12 hours after the patient is first seen will provide a pattern to determine whether the CK-MB is rising, indicative of myocardial injury. The CK-MB is also useful for diagnosis of reinfarction or extensive of an MI because it begins to fall after a day, dissipating in 1 to 3 days, so subsequent elevations are indicative of another event.
A 'cardiac index' can provide a useful indicator for early MI. This is calculated as a ratio of total CK to CK-MB, and is a sensitive indicator of myocardial injury when the CK-MB is elevated.
CK-MB Isoforms The CK-MB fraction exists in two isoforms called 1 and 2 identified by electrophoretic methodology. The ratio of isoform 2 to 1 can provide information about myocardial injury.
An isoform ratio of 1.5 or greater is an excellent indicator for early acute myocardial infarction. CK-MB isoform 2 demonstrates elevation even before CK-MB by laboratory testing. However, the disadvantage of this method is that it is skilled labor intensive because electrophoresis is required, and large numbers of samples cannot be run simultaneously nor continuously. False positive results with congestive heart failure and other conditions can occur.
Troponins: Troponin I and T are structural components of cardiac muscle. They are released into the bloodstream with myocardial injury. They are highly specific for myocardial injury--more so than CK-MB--and help to exclude elevations of CK with skeletal muscle trauma. Troponins will begin to increase following MI within 3 to 12 hours, about the same time frame as CK-MB. However, the rate of rise for early infarction may not be as dramatic as for CK-MB.
Troponins will remain elevated longer than CK--up to 5 to 9 days for troponin I and up to 2 weeks for troponin T. This makes troponins a superior marker for diagnosing myocardial infarction in the recent past--better than lactate dehydrogenase (LDH). However, this continued elevation has the disadvantage of making it more difficult to diagnose reinfarction or extension of infarction in a patient who has already suffered an initial MI. Troponin T lacks some specificity because elevations can appear with skeletal myopathies and with renal failure.
Myoglobin: Myoglobin is a protein found in skeletal and cardiac muscle which binds oxygen. It is a very sensitive indicator of muscle injury. The rise in myoglobin can help to determine the size of an infarction. A negative myoglobin can help to rule out myocardial infarction.. It is elevated even before CK-MB. However, it is not specific for cardiac muscle, and can be elevated with any form of injury to skeletal muscle.
Lactate dehydrogenase The LDH has been supplanted by other tests. It begins to rise in 12 to 24 hours following MI, and peaks in 2 to 3 days, gradually dissipating in 5 to 14 days. Measurement of LDH isoenzymes is necessary for greater specificity for cardiac injury. There are 5 isoenzymes (1 through 5). Ordinarily, isoenzyme 2 is greater than 1, but with myocardial injury, this pattern is 'flipped' and 1 is higher than 2. LDH-5 from liver may be increased with centrilobular necrosis from passive congestion with congestive heart failure following ischemic myocardial injury.
The next table shows the time courses of cardiac enzymes.
Time Course of Cardiac Enzymes Following Acute
|
Enzyme |
Onset |
Peak |
Return to normal |
|
CK CK-MB LDH LDH1 LDH2 |
3 6 hr 2 4 hr 24 hr 4 hr 4 hr |
12 24 hr 12 20 hr 48 72 hr 48 hr 48 hr |
3 5 days 48 72 hr 7 10 days 10 days 10 days |
3.6. Medical Management
The goal medical management is to minimize myocardial damage. Minimizing myocardial damage is accomplished by relieving pain, providing rest, and preventing complications such as lethal dysrhythmias and cardiogenic shock.
The most critical period for the patient with an MI is the first 2 to 3 days after the attack. The area of infarction can increase in size for several hours or days after the onset of the attack. Cardiogenic shock and ventricular fibrillation are common causes of sudden death during this period.
Thrombolytic drugs are contraindicated for the treatment of unstable angina and NSTEMI and for the treatment of individuals with evidence of cardiogenic shock.
Although no perfect thrombolytic agent exists, an ideal thrombolytic drug would lead to rapid reperfusion, have a high sustained patency rate, be specific for recent thrombi, be easily and rapidly administered, create a low risk for intra-cerebral and systemic bleeding, have no antigenicity, adverse hemodynamic effects, or clinically significant drug interactions, and be cost effective. Currently available thrombolytic agents include streptokinase, urokinase, and anistreplase (recombinant tissue plasminogen activator, t-PA). More recently, thrombolytic agents similar in structure to t-PA such as reteplase and tenecteplase have been used. These newer agents boast efficacy at least as good as rtPA with significantly easier administration. The thrombolytic agent used in a particular individual is based on institution preference and the age of the patient.
Depending on the thrombolytic agent being used, adjuvant anticoagulation with heparin or low molecular weight heparin may be of benefit. With t-PA and related agents (reteplase and tenecteplase), heparin is needed to maintain coronary artery patency. Because of the anticoagulant effect of fibrinogen depletion with streptokinase and urokinase treatment, it is less necessary there.
Intracranial bleeding (ICB) and subsequent cerebrovascular accident (CVA) is a serious side effect of thrombolytic use. The risk of ICB is dependent on a number of factors, including a previous episode of intracranial bleed, age of the individual, and the
thrombolytic regimen that is being used. In general, the risk of ICB due to thrombolytic use for the treatment of an acute myocardial infarction is between 0.5 and 1 percent.
Thrombolytic therapy to abort a myocardial infarction is not always effective. The degree of effectiveness of a thrombolytic agent is dependent on the time since the myocardial infarction began, with the best results occurring if the thrombolytic agent is used within two hours of the onset of symptoms. If the individual presents more than 12 hours after symptoms commenced, the risk of intracranial bleed are considered higher than the benefits of the thrombolytic agent. Failure rates of thrombolytics can be as high as 20% or higher. In cases of failure of the thrombolytic agent to open the infarct-related coronary artery, the patient is then either treated conservatively with anticoagulants and allowed to 'complete the infarction' or percutaneous coronary intervention (PCI, see below) is then performed. Percutaneous coronary intervention in this setting is known as 'rescue PCI' or 'salvage PCI'. Complications, particularly bleeding, are significantly higher with rescue PCI than with primary PCI due to the action of the thrombolytic agent.
Bibliography:
Braunnwald
E. Heart Disease: A textbook of Cardiovascular Medicine.
Brunner and Suddarth s Textbook of Medical Surgical Nursing, Seventh Edition
|
Politica de confidentialitate | Termeni si conditii de utilizare |

Vizualizari: 1508
Importanta: ![]()
Termeni si conditii de utilizare | Contact
© SCRIGROUP 2025 . All rights reserved