| CATEGORII DOCUMENTE |
| Bulgara | Ceha slovaca | Croata | Engleza | Estona | Finlandeza | Franceza |
| Germana | Italiana | Letona | Lituaniana | Maghiara | Olandeza | Poloneza |
| Sarba | Slovena | Spaniola | Suedeza | Turca | Ucraineana |
Full Table of Contents
Esters, Organic
1. Introduction
2. Physical Properties
3. Chemical Properties
3.1. Hydrolysis
3.2. Transesterification
3.3. Ammonolysis
3.4. Reduction
3.5. Claisen Condensation
3.6. Pyrolysis
4. Natural Sources
5. Production
5.1. Esterification of Carboxylic Acids
5.1.1. Catalysts
5.1.2. Industrial Processes
5.2. Alkylation of Metal Carboxylates
5.3. Acylation with Acyl Halides
5.4. Acylation with Carboxylic Anhydrides
5.5. Transesterification
5.6. Acylation with Ketenes
5.7. Carbonylation
5.8. Condensation of Aldehydes
5.9. Alcoholysis of Nitriles
5.10. Acylation of Olefins
5.11. Ethoxylation of Carboxylic Acids
5.12. Orthocarboxylic Acid Esters
5.13. Lactones
6. Environmental Protection
7. Quality Specifications and Analysis
8. Storage and Transportation
9. Uses and Economic Aspects
10. Toxicology and Occupational Health
11. References
Table 1 - Physical properties of the most important esters
Table 2 - Water solubility of lower esters
Table 3 - Relative rates of acid-catalyzed hydrolysis of esters
Table 4 - Composition of common azeotropes
Table 5 - Biodegradation of organic esters
Table 6 - Toxicity for fishes and bacteria
Table 7 - Typical specifications of commercial esters
Table 8 - Production of miscellaneous esters in 1985, t
Table 9 - Production of plasticizers in 1985, t
Table 10 - United States production of esters used as surfactants (1985)
Table 11 - United States production of flavor and perfume esters (1985)
Table 12 - Toxicity data for various esters
1. Introduction
Organic esters are compounds with the general formula

where R1 and R2 represent either the same or different aliphatic, aromatic, or heterocyclic groups. Esters are classified as carboxylic acid derivatives; they are prepared mainly by the reaction of a carboxylic acid and an alcohol (esterification).
Ortho esters are derived from ortho acids:
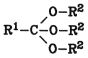
Ortho esters can be hydrolyzed to carboxylic acids and alcohols, but their properties are more similar to those of acetals. They are dealt with briefly in Section 5.12. Orthocarboxylic Acid Esters
If a molecule possesses both a carboxyl and a hydroxyl group, it may form either polyesters:
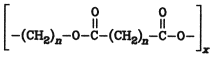
or cyclic internal esters (lactones) (Section 5.13. Lactones
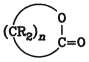
Ring opening reactions of lactones lead to the same type of polyesters. Another type of polyesters with different characteristics is usually produced through condensation of dicarboxylic or polycarboxylic acids with di- or polyhydric alcohols.
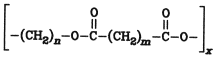
Organic esters are of considerable economic importance. Esters of glycerol with fatty acids (glycerides) occur naturally in large quantities in fats and fatty oils (see Chap. 4. Natural Sources and Fats and Fatty Oils); they are used predominantly in foods, but also as raw materials, especially in the production of surfactants ( Surfactants Laundry Detergents; Soaps Fatty Acids Fatty Alcohols
Cellulose acetate is used on a large scale in fiber production. Synthetic esters are of increasing importance in many applications, e.g., fibers, films, adhesives, and plastics articles. Various synthetic esters have been developed for specific uses, e.g., solvents, extractants, plasticizers, lubricants, lubricant additives, and lacquer bases (see also Solvents Plasticizers). A variety of volatile esters are used as aromatic materials in perfumes, cosmetics, and foods ( Flavors and Fragrances
This article gives a general survey of production methods, properties, and uses of esters. The commercially most important esters are treated under separate keywords: Formic Acid Acetic Acid Propionic Acid and Derivatives Adipic Acid Acrylic Acid and Derivatives Methacrylic Acid and Derivatives Benzoic Acid Phthalic Acid and Derivatives Terephthalic Acid, Dimethyl Terephthalate, and Isophthalic Acid Fats and Fatty Oils Waxes Cellulose Esters Polyesters
Nomenclature. The term ester was coined by gmelin (1850) from the German word Essig-ther (acetic ether, ethyl acetate). According to IUPAC rule C463, esters of carboxylic acids are named by substituting the ending -ate for the -ic acid; the alkyl or aryl group is cited first. For example, the methyl ester of propionic acid is called methyl propionate . Alternatively, the term propionic acid methyl ester is used.
Acid esters, such as the monoesters of dibasic acids, are named by inserting the word hydrogen between the names of the alcohol and acid (IUPAC rule C463.2). Thus, methyl hydrogen succinate is the monomethyl ester of succinic acid.
Esters of glycerol such as fats and fatty oils are known as glycerides. Trivial names are usually preferred:

Ortho esters are named as derivatives of ortho acids, e.g., trimethyl orthoacetate (IUPAC rule C464).
In substitutive nomenclature, different prefixes are used for the ester group, depending on the position of the principal group R:
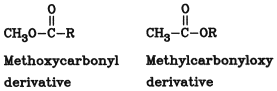
J. D. Fletcher, O. C. Dermer, R. B. Fox (eds.): Nomenclature of Organic Compounds, Am. Chem. Soc., Washington D.C. 1974, pp. 137 145.IUPAC commission on nomenclature of organic chemistry: Nomenclature of Organic Chemistry, 4th ed., Pergamon Press, Oxford-New York 1979.
2. Physical Properties
The physical properties of commercially important aliphatic and aromatic esters are listed in Table . The lower esters are colorless, mobile, and highly volatile liquids that usually have pleasant odors. As the molecular mass increases, volatility decreases and the consistency becomes waxy, then solid, and eventually even brittle, often with formation of lustrous crystals.
The melting point of an ester is generally lower than that of the corresponding carboxylic acid. The boiling point depends on the chain length of the alcohol component and may eventually exceed that of the acid. The lower esters are relatively stable when dry and can be distilled without decomposition.
With the exception of low molecular mass compounds formed by short-chain carboxylic acids, esters are insoluble in water (Table ). By contrast, they are readily miscible with many organic solvents.
K. S. Markley (ed.): Fatty Acids, Their Chemistry, Properties, Production and Uses, 2nd ed., Interscience, New York 1960/61.
3. Chemical Properties
Due to the large number of possible acid and alcohol moieties, the chemical properties of esters may differ considerably. Only typical reactions applicable to the majority of esters are described in the following sections.
3.1. Hydrolysis
Esters are gradually split into the acid and alcohol through the action of humidity:

This hydrolysis is catalyzed by acids (protons) or bases (hydroxyl ions). The catalytic effect of bases is generally stronger. Industrially, the hydrolysis of esters is usually carried out with bases. However, acid hydrolysis is preferred for the production of pure acids and for alkali-sensitive compounds.
Hydrolysis of esters by use of water and a mineral acid leads to an equilibrium mixture of ester, alcohol, and free carboxylic acid. If bases are used, the salt of the carboxylic acid is formed; the carboxylic acid is thus removed from the equilibrium and the reaction becomes irreversible:
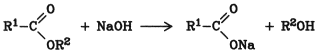
Higher temperatures accelerate the reaction. Table gives the relative rates of hydrolysis for some esters: hydrolysis is hindered by bulky acyl or alcohol groups, whereas acidifying groups increase the reaction rate.
Esters of secondary alcohols are hydrolyzed more slowly than those of primary alcohols. Esters of tertiary alcohols, e.g., tert-butyl esters, are exceptional in that they are readily hydrolyzed in the presence of acids and then often yield a-olefins instead of alcohols. Some esters are hydrolyzed only under extreme conditions; for example, esters of pivalic acid [75-98-9] (2,2-dimethylpropionic acid) and ortho-substituted benzoic acids react only in concentrated H2SO4.
Basic Hydrolysis. Basic hydrolysis (saponification) is one of the earliest known chemical reactions. By this method, soaps are obtained from natural fats and oils, with glycerol as a byproduct. Saponification based on wood ash as the alkali was described as early as 2500 bc; the principle is still employed today ( Soaps). The reaction was investigated by scheele in 1779.
Basic hydrolysis requires stoichiometric amounts of alkali. The reaction is carried out by boiling the ester in an aqueous alkaline solution. Under these conditions, the alkali salt of the carboxylic acid is dissolved.
Acidic Hydrolysis. If the goal is to obtain free carboxylic acids rather than soaps, acid hydrolysis is the preferred method. Free acids are needed for various industrial purposes ( Carboxylic Acids, Aliphatic Fatty Acids). Complete hydrolysis can only be achieved by removal of the alcohol from the equilibrium. The reaction rate in dilute acids is generally fairly low, mainly because of the poor solubility of esters in water. Therefore, in practice, emulsifiers such as sulfonated oleic acid or sulfonated aromatic compounds (Twitchell reagent) are added and the reaction is carried out at ca. 100 C under pressure.
Another industrial process involves high-temperature hydrolysis with steam at 170 300 C under pressure. Zinc, calcium, and magnesium compounds are used as catalysts . Vinyl esters are readily hydrolyzed in the presence of palladium salts
Enzymatic Hydrolysis. Specific lipolytic enzymes, the so-called esterases, effect hydrolysis at temperatures below 40 C. Thus, lipase occurs in the digestive tracts of humans and most animals and in plants. Castor oil lipase is of commercial importance in hydrolysis of fats under mild conditions. The enzymes are especially effective in the weakly acidic range. Lipases may achieve importance as detergent additives; at low temperatures (20 C) they facilitate the removal of fat-containing soils ( Laundry Detergents - 3.4.1. Enzymes
D. Swern (ed.): Bailey's Industrial Oil and Fat Products, Wiley-Interscience, New York 1969, p. 937.
J. Smidt et al., Angew Chem. 74 (1962) 93.
3.2. Transesterification
When esters are heated with alcohols, acids, or other esters, the alcohol or acid groups are exchanged more or less completely. This process is called transesterification. It is accelerated in the presence of a small amount of acid or alkali.
Three types of transesterification are known:
Exchange of alcohol groups (alcoholysis)

Exchange of acid groups (acidolysis)
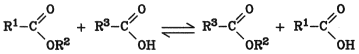
Ester ester interchange
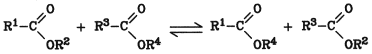
All three are equilibrium reactions and proceed to completion only if one component is removed from the reaction mixture.
Dispersed alkali metals, mainly sodium, are suitable catalysts for the transesterification of fats . Tin salts have been used in the production of aryl esters, e.g., phenyl esters, at 190 250 C. Methacrylic acid esters of higher alcohols, e.g., ethylhexanol, can be obtained by transesterification in the presence of tin compounds or with magnesium alkoxides . Other catalysts are discussed in Section 5.5. Transesterification
Transesterification reactions play a major role in industry and are important in laboratory practice and in analytical chemistry. They can be used to reduce the boiling point of esters by exchanging a long-chain alcohol group for a short one such as methanol, e.g., in the analysis of fats, oils, and waxes (see Chap. 7. Quality Specifications and Analysis) and in vapor-phase reactions. Transesterification reactions can also be useful if direct esterification with the desired alcohol is technically difficult because of physical reasons (e.g., high boiling point, low solu-bility, or high viscosity). Examples are to be found in polymer chemistry. Thus, poly(ethylene terephthalate) is produced mainly by transesterification of dimethyl terephthalate with ethylene glycol. Propanediols and 1,4-butanediol are used in a similar manner. The reaction is catalyzed with calcium, antimony, titanium, and germani-um salts. A further example is the reaction of diols (e.g., bisphenol A) with carbonic acid esters to produce polycarbonates.
An industrial example of acidolysis is the reaction of poly(vinyl acetate) with butyric acid to form poly(vinyl butyrate). Often a butyric acid methanol mixture is used and methyl acetate is obtained as a coproduct.
Unilever, DE 2 327 729, 1973 (J. J. Muller, T. J. Kock).
Rohm & Haas, DE 1 932 625, 1969 (W. R. Winslow).
Degussa, DE 1 568 376, 1966 (G. Morlock, H. Trageser).
3.3. Ammonolysis
Ammonia and esters react to form alcohols and amides:
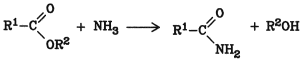
This reaction is similar to hydrolysis. It is carried out in aqueous or alcoholic ammonia. Lower esters give good yields even at room temperature; higher esters require higher temperature and pressure.
If primary or secondary amines are used, N-substituted amides are formed. This reaction is called aminolysis. Hydrazines yield the corresponding hydrazides.
If esters are passed with ammonia over a catalyst such as alumina at 400 500 C, dehydration of the intermediate amides occurs and the corresponding nitriles are obtained directly.

Fatty nitriles can be produced in this manner (see also Amines, Aliphatic - 7.2. Production
A. M. Schwartz, J. W. Perry, J. Berch: Surface-Active Agents and Detergents, vol. 2, Interscience, New York 1958, p. 104.
3.4. Reduction
Under certain conditions esters can be reduced to alcohols.
Bouveault Blanc Reduction. This classical method developed in 1904 uses metallic sodium in alcohol ( Fatty Alcohols). However, the Bouveault Blanc reduction has been replaced nearly completely by catalytic hydrogenation and remains important only for laboratory purposes. The advantage of the Bouveault Blanc process is that only the ester group is hydrogenated, while carbon carbon double bonds are not reduced. Therefore, the reaction is suitable for the synthesis of unsaturated alcohols such as oleyl alcohol from unsaturated esters.
Catalytic Hydrogenation. A process of great industrial importance is the catalytic reduction of esters with hydrogen at 200 300 C and 10 30 MPa (100 300 bar) in the presence of copper chromite catalysts

Sulfur and halogens are catalyst poisons. This method gives high yields and is used for the large-scale reduction of fats and fatty oils such as coconut oil and tallow, usually after transesterification to the methyl esters ( Fatty Alcohols). The main products are dodecyl and decyl alcohols, which are important raw materials for the production of detergents. The method is also suitable for the conversion of 1,4-dimethyl terephthalate into 1,4-bis(hydroxymethyl) cyclohexane [105-08-8] , an important intermediate in the production of polyesters.
Hardening of fats (hydrogenation of double bonds in unsaturated fats and oils) is carried out in the presence of nickel catalysts under slight hydrogen pressure and does not involve ester splitting.
Grignard Reaction. Esters react with alkylmagnesium halides under Grignard conditions. Formic acid esters give secondary alcohols; the other esters yield tertiary alcohols.
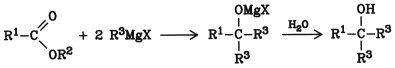
Yields are high, e.g., 95 % in the production of triphenylmethanol from ethyl benzoate and phenylmagnesium bromide. The method is used mainly on a laboratory scale.
Reduction with Complex Metal Hydrides. The reduction of esters with complex metal hydrides, especially lithium aluminum hydride, has only preparative impor-tance . Its advantage is its broad applicability for all types of esters; double bonds are preserved. This method leads to very pure alcohols.
H. Adkins, Org. React. (N.Y.) 8 (1954) 1
N. G. Gaylord: Reduction with Complex Metal Hydrides, New York 1956, pp. 391
3.5. Claisen Condensation
The reaction of carboxylic acid esters with reactive methylene and methyl groups in the presence of alkaline compounds such as sodium alkoxides leads to the formation of b oxocarboxylic acid esters (b-keto esters) . The classical example is the condensation of two molecules of ethyl acetate to form acetoacetate:
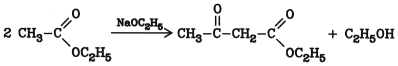
Oxocarboxylic acid esters are among the most important intermediates in the laboratory and in industry. During hydrolysis with dilute alkali, these esters readily decompose into ketones, alcohols, and carbon dioxide:
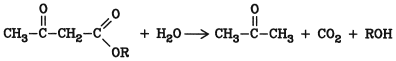
Almost any ketone can be synthesized by using this process.
In industry, the Claisen condensation has been replaced by the synthesis of acetoacetic acid derivatives from diketene [674-82-8] , see also Section 5.6. Acylation with Ketenes Oxocarboxylic Acids, Aliphatic Ketenes
Condensation of esters in inert solvents in the presence of sodium rather than sodium ethoxide leads to formation of diketones and a-hydroxy ketones (acyloins) . This reaction is utilized in the synthesis of large-ring compounds.
C. B. Hauser, B. E. Hudson, Jr., Org. React. (N.Y.) 1 (1947) 266, 302.
J. J. Bloomfield et al., Org. React. (N.Y.) 23 (1976) 259
3.6. Pyrolysis
When passed at 300 500 C over an inert heat-transfer agent such as quartz glass, esters decompose into carboxylic acid and the unsaturated compound that corresponds to the alcohol. Esters of primary alcohols generally lead to a-olefins:

The double bond does not migrate along the carbon chain. Methyl esters and other esters without hydrogen atoms in the b-position do not yield uniform pyrolysis products.
When heated to high temperatures in the presence of metal oxides such as thorium oxide, calcium oxide, manganese chromite, or zinc chromite, fatty acid esters or free fatty acids give high yields of ketones:
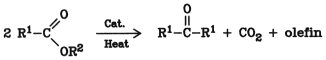
The alcohol moiety is split off as olefin.
4. Natural Sources
Esters occur naturally in large quantities in fats and fatty oils, waxes, and fruit ethers.
Fats and Fatty Oils . Fats and fatty oils (as opposed to mineral oils) are almost exclusively esters of glycerol with long- and medium-chain fatty acids. About 1300 different fats and oils are known. They contain mainly even-numbered, straight-chain carboxylic acids ( Fats and Fatty Oils). The most common are the triglycerides of stearic, palmitic, and oleic acid. The melting point of the natural products varies between 55 C (mutton tallow) and 27 C (nut oil) and decreases as the content of unsaturated and short-chain fatty acids increases.
Only a few of the animal fats and vegetable oils are suitable for commercial production. They are predominantly used as foods, but have also gained considerable industrial importance, e.g., in the production of lacquers, detergents, and solvents. The most frequently produced fats of animal origin are butter, beef and mutton tallow, lard, fish oil, and whale oil. Butter accounts for ca. 40 % of the world production of animal fats. The most important oils of plant origin are soybean oil, sunflowerseed oil, peanut oil, cottonseed oil, coconut oil, rapeseed oil, palm oil, and olive oil. Linseed oil, hempseed oil, and poppyseed oil contain a variable amount of unsaturated fatty acids, e.g., linoleic acid, linolenic acid, ricinoleic acid, and erucic acid. These oils gradually harden under the influence of oxygen in the air ( Drying Oils and Related Products
Waxes. Natural waxes (wax esters) are esters of monobasic carboxylic acids with higher monohydric and, less commonly, dihydric alcohols. Currently they are largely replaced by synthetic waxes, which are in most cases long-chain hydrocarbons, but not esters ( Waxes
The most important sources of wax esters are beeswax, whose main constituent is myricyl palmitate [6027-71-0]; spermaceti from the head oil of the sperm whale, which contains cetyl palmitate [540-10-3]; and carnauba wax from the Brazilian palm, containing myricyl cerotate [84324-99-2]. Fossil waxes, such as montan waxes in bituminous lignite and peat waxes, also have major wax ester components. Lanolin, the neutral part of wool grease, contains higher fatty acid esters of cholesterol.
Fruit Ethers. Fruit ethers, i.e., volatile esters of short- and medium-chain carboxylic acids with short- and medium-chain alcohols, are major constituents of essential oils. Their name derives from their pleasant, usually fruity odor. They are used as aromatic materials in fragrances, foods, beverages, cosmetics, and soaps ( Flavors and Fragrances). Natural fruit ethers are obtained from plant materials by extraction, steam distillation, pressing, or a combination of these methods. The majority of simple, short-chain esters are currently produced synthetically.
5. Production
A great variety of production methods for carboxylic acid esters are known. However, many of these methods are valuable only for preparing special compounds . In what follows, only those methods that have industrial significance are discussed.
Houben-Weyl, E5, 656
5.1. Esterification of Carboxylic Acids
The simplest and most common method of esterification is the reaction of a concentrated alcohol with a concentrated carboxylic acid with the elimination of water:
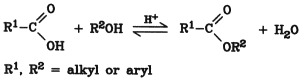
Esterification is the reverse of hydrolysis and leads to an equilibrium (see Section 3.1. Hydrolysis). With acetic acid and ethanol, the molar equilibrium ratio of ethyl acetate to acetic acid is ca. 2 1 (65 mol % of ethyl acetate). The equilibrium may be shifted toward the ester by increasing the concentration of one of the reactants, usually the alcohol. However, quantitative esterification is possible only by continuous removal of one of the products, i.e., ester or water. Usually the water is distilled off as an azeotrope (see Section 5.1.2. Industrial Processes
Removal of the water by chemical or adsorptive binding is also possible. In preparative chemistry, calcium carbide, calcium hydride, and calcium and magnesium sulfate have proved successful; in industrial applications, activated bauxite is also used. The distilled, water-rich condensates are passed over the water-binding agents; alternatively, salts such as copper sulfate or calcium chloride can be added directly to the reaction mixture. Molecular sieves are also suitable . Acetone dimethyl acetal, which binds water and simultaneously supplies methyl groups, may be used for the production of methyl esters
Esterification is carried out at high temperature and in the presence of catalysts. The rate of ester formation depends on the carboxylic acid and the alcohol used. The lowest members, i.e., methanol and formic acid, react the most readily. Primary alcohols react faster than secondary alcohols, and the latter react faster than tertiary ones. Within each series, the reaction rate generally decreases with increasing molecular mass. Straight-chain acids react more readily than branched ones; the rate of esterification is lowered particularly by branching in the a-position. The relative rates of esterification are similar to those of hydrolysis (see Table ). Esterification of aromatic acids, e.g., benzoic acid, is slow.
Experiments with primary and secondary alcohols containing 18O have shown that the oxygen in the water formed during acid-catalyzed esterification originates from the acid, not the alcohol . The reaction of tertiary alcohols is more complex and does not proceed clearly via a tertiary carbonium ion. The mechanism is discussed in
5.1.1. Catalysts
Mineral Acids. Only strong carboxylic acids react sufficiently quickly without a catalyst. Generally, a strong mineral acid must be added. Suitable catalysts include sulfuric acid, hydrogen chloride, arylsulfonic acids such as p-toluenesulfonic acid, and chlorosulfuric acid. Phosphoric acid, polyphosphoric acids, and mixtures of acids are also recommended . If the acids are adsorbed on a solid support, esterification can be carried out as a continuous process.
Ion Exchangers. The acid groups can be bound chemically to a polymeric material. Cat-ion exchangers such as sulfonated polystyrene permit esterification under mild reaction conditions. The resulting esters are generally very pure because acid-catalyzed side reactions such as dehydration, etherification, and rearrangement are almost completely suppressed . This method gives high yields and is widely used in industry. After the reaction water has been removed, the ion exchanger is filtered off and the ester is purified by distillation. Zeolites are also used as esterification catalysts
Lewis Acids. Lewis acids such as boron trifluoride increase the reactivity of the carboxyl group toward alcohols. Boron trifluoride reacts with alcohols under proton release:
BF3 + ROH [ BF3OR] + H+
The proton acts as a catalyst. The complex with diethyl ether, boron trifluoride etherate [109-63-7] , is generally preferred because it is easier to handle than gaseous boron trifluoride . This method of esterification proceeds under mild conditions and is widely applicable. However, it is limited to the manufacture of high-value esters because of the relatively high price of BF3. Each carboxyl group requires one equivalent of the boron trifluoride complex, which in most cases is destroyed during the further processing of the ester. All other groups that possess a free electron pair (e.g., hydroxyl, oxo, or amino groups) require an additional equivalent of BF3.
Metal salts may also function as Lewis acids. For example, a complex acid is formed from the reaction of a zinc salt and an alcohol:

The actual catalysts are again the protons. Tin compounds have been used to prepare glyceryl esters. For the production of dimethyl ter-ephthalate, silica treated with aluminum, titanium, zinc, or tin compounds has proved successful ; this esterification proceeds also in the absence of catalysts (12.5 14.0 MPa, 270 275 C, 95 % yield).
Amphoteric Hydroxides. Esterification is catalyzed by amphoteric hydroxides of metals of groups I, II, III, and IV, especially aluminum hydroxide. The actual catalyst is probably the aluminate anion:
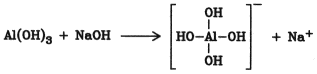
The aluminate may be generated in the reaction mixture from aluminum hydroxide and sodium hydroxide . The esters produced by this method are light in color, and purification by distillation is generally unnecessary. Therefore, this process is especially suitable for preparing esters with high boiling points. Its main application is in the production of plasticizers, e.g., phthalates.
Other Compounds. A possible esterification catalyst is graphite hydrogen sulfate [12689-13-3] , (C24HSO4 2 H2SO4)57. It is obtained by electrolysis of 98 % sulfuric acid with a graphite anode.
Silanes and phosphoric acid derivatives are utilized for syntheses under mild conditions, the latter especially for phenyl carboxylates.
Acid esterification of amino acids and peptides is difficult because protecting groups and peptide bonds are easily hydrolyzed. High yields of esters of both protected and free amino acids or peptides are obtained in the presence of a mild dehydration catalyst such as sulfuryl chloride or thionyl chloride at room temperature
Trifluoroacetic anhydride is a suitable catalyst for the esterification of sterically hindered acids
5.1.2. Industrial Processes
Esterification is generally carried out by refluxing the reaction mixture until all the water has been split off. The water or the ester is removed from the equilibrium by distillation. The choice of method to achieve complete esterification depends on the boiling points of the alcohol, the acid, and the ester.
Basically, the following three possibilities exist:
The boiling point of the ester is lower than that of water. In this case the ester can be distilled off together with the alcohol. This method is used to produce methyl acetate, which forms an azeotrope with methanol.
Ester and water can be distilled off together, usually as an azeotropic mixture. On condensation, the mixture separates into an ester and a water phase. To achieve complete distillation of the ester, water or steam are often added to the reaction mixture. An example of this method is the production of sec-butyl acetate. Frequently alcohol, ester, and water form ternary azeotropes, e.g., during the esterification of acetic acid with ethanol.
The boiling point of the ester is higher than that of water. In this case, the water is distilled off, frequently as an azeotrope with the alcohol. Except for methanol, all alcohols of medium volatility form azeotropes with water. If the alcohol water mixture separates into two phases during condensation, the alcohol can be recycled into the reaction mixture , as exem-plified in the production of n-dibutyl phthalate. If water and alcohol do not separate, fresh alcohol must be continuously introduced into the reaction mixture.
Removal of water usually involves the addition of entrainers, which form azeotropes with relatively low boiling points and high water content. Table lists some frequently used entrainers. After condensation, the azeotrope separates into an aqueous phase and an organic phase, and the entrainer is recycled into the reaction mixture.
With ethanol, n-propanol, 2-propanol, allyl alcohol, and tert-butyl alcohol, a cosolvent such as benzene or toluene is often added to the condensate to achieve separation of the organic phase
With high-boiling esters, e.g., esters of polyhydric alcohols, the water of reaction may be removed by means of steam or inert gases. Steam distillation can also be used to remove excess alcohol after the esterification is complete.
Esterification is usually carried out batchwise. In commercial production, continuous processes are preferred. Special procedures have been devised in some cases.
Methyl and ethyl esters of mono- and dicarboxylic acids are produced continuously by pumping the alcohol and the carboxylic acid melt (e.g., lauric acid or adipic acid) into a pipe from below and drawing off the ester at the top . In a continuous process for the production of volatile esters, the mixture of acid, alcohol, and catalyst is introduced into the upper part of a distillation column and an excess of the alcohol is introduced into the bottom. The column is heated so that the ester, water, and excess alcohol are distilled off
Nonvolatile esters such as bis(2-ethylhexyl) phthalate [117-81-7] can be obtained continuously by the following procedure:
The mixture of acid or anhydride and alcohol together with sulfuric acid is introduced at the top of a plate column. An entrainer, e.g., benzene, is distilled through the column from below. The azeotrope of water and entrainer is drawn off the top and the ester accumulates in the sump.
Monochloroacetic acid esters of alcohols with three to five carbon atoms are obtained continuously by adding equimolar amounts of chloroacetic acid and alcohol to a chloroacetic acid melt and distilling off the ester and water.
Esterification can also be carried out continuously in the vapor phase by heating a mixture of acid, alcohol, ester, and catalyst to the reaction temperature. This process is called gas-phase esterification although one or more of the components may be present as liquids or solids.
Ion-exchange resins are especially suitable as catalysts in continuous processes. The reactants pass through or over the solid catalyst, and no separation or neutralization of the catalyst is necessary
H. R. Harrison, W. M. Heynes et al., Chem. Ind. (London) 1968, 1568.
J. R. Rachele, J. Org. Chem. 28 (1963) 2898.
I. Roberts, H. C. Urey, J. Am. Chem. Soc. 60 (1938) 2391.
H. Zimmermann, J. Rudolf, Angew. Chem. 77 (1965) 65 74;Angew. Chem. Int. Ed. Engl. 4 (1965) 40.
R. Bader, A. D. Kontowicz, J. Am. Chem. Soc. 75 (1953) 5116.
Knapsack, DE 2 226 829, 1972 (E. Lohmar, F. X. Werber).
G. A. Olah et al., Synthesis 1978, 929 ff.
H. J. Bergk, Z. Chem. 18 (1978) 22 ff.
G. Hallas, J. Chem. Soc. 1965, 5770.
B. F. Goodrich Comp.DE 1 005 947, 1955 (W. L. Bears, F. X. Werber).B. F. Goodrich Comp., DE 1 103 335, 1956 (F. X. Werber, S. J. Averill).Reichhold Chemie AG, DE 1 173 473, 1962 (H. Dalibar).Reichhold Chemie AG, DE-AS 1 185 611, 1963 (H. Dalibar).ICI, DE-AS 1 292 140, 1966 (D. K. Steel).Scholven Chemie AG, DE-OS 1 593 503, 1966 (K. S. Schmitt, W. Flakus et al.).Mobil Oil Comp., DE-AS 1 618 639, 1967 (A. B. Gainor, L. McMakin)British Titan Products Comp., DE-OS 1 807 103, 1968 (R. H. Stanley, D. W. Brook, et al.).Hls AG, DE-AS 2 243 240, 1972 (F. List, K. Wember)F. Brill, L. Ferry, I. T. Baker, High Polym. 27 (1972) 506.
Hls, DE 914 006, 1951 (A. Coenen, F. Broich).
E. Taschner, C. Wasielewski, Liebigs Ann. Chem. 640 (1961) 136
R. C. Parish, C. M. Stock, J. Org. Chem. 30 (1965) 927.
S. Natelson, S. Gottfried, Org. Synth., Coll. Vol. 3 (1955) 381.
Ruhrchemie, DE 1 171 897, 1959 (H. Feichtinger, H. Noerke).
BASF, DE 878 348, 1942 (H. Dierichs, E. Braun, et al.).
5.2. Alkylation of Metal Carboxylates
If metal carboxylates are treated with alky-lating agents, good yields of carboxylic acid esters result. A one-pot procedure is often used, in which the salt is first produced from the carboxylic acid and then directly alkylated to form the ester. Isolation of the salt is necessary only in the case of thermally unstable quaternary ammonium salts
Alkali metal and alkaline-earth metal salts, as well as silver and copper salts, can be used. Dialkyl sulfates, aliphatic halides such as long-chain alkyl chlorides, and alicyclic halides are suitable alkylating agents. The method is used mainly in preparative chemistry.
W. William, B. Halper, Synthesis 1974, 727.
5.3. Acylation with Acyl Halides
High yields of esters are obtained by acylation of alcohols with carboxylic acid halides:

The acyl chloride is often produced in situ. The liberated hydrogen halide is generally bound to a base, e.g., pyridine or alkali hydroxide (Schotten Baumann reaction).
This method is particularly important in preparative and analytical chemistry. A commercial application is the production of polycarbonates from phosgene and diols. Phenyl esters and esters of sterically hindered carboxylic acids can be produced from the corresponding acyl halides in the presence of Lewis acids, e.g., iron(III), aluminum, tin(IV), and titanium(IV) chlorides. Addition of phase-transfer agents such as tetrabutylammonium chloride has also proved effective . An important application of this method is in the esterification of steroids.
G. Schwabe, G. Westphal, H. G. Hennig, Z. Chem. 20 (1980) 184.S. Szeja, Synthesis 1980, 402.
5.4. Acylation with Carboxylic Anhydrides
Carboxylic anhydrides are suitable acylating agents for alcohols and phenols. With the exception of trifluoroacetic anhydride and mixed carboxylic anhydrides, they are less reactive than acyl halides.
The advantage of this method is that no water is formed; however, only one equivalent acyl per molecule of anhydride is available for esterification:
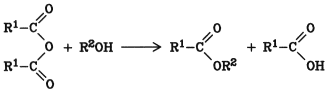
Therefore, the method is especially suitable for the esterification of rare or expensive alcohols with cheap anhydrides such as acetic anhydride.
Acylation can be accelerated by addition of protonic acids (sulfuric acid, hydrochloric acid, perchloric acid, p-toluenesulfonic acid) or Lewis acids (zinc chloride). A large-scale commercial process is the production of cellulose acetate from cellulose and acetic anhydride ( Cellulose Esters - 2.1. Cellulose Acetate
Another important product is acetylsalicylic acid (aspirin), which is obtained from salicylic acid and acetic anhydride.
Base-catalyzed acylation with anhydrides is used to synthesize esters from sterically hindered or acid-sensitive alcohols . Common bases are triethylamine and pyridine.
Reaction with anhydrides is the basis of a quantitative analytical method for hydroxyl groups.
J. Cason, Org. Synth., Coll. Vol. 3 (1955) 169.
W. Steglich, DE 1 958 954, 1969 (W. Steglich, G. Hfle).
5.2. Alkylation of Metal Carboxylates
If metal carboxylates are treated with alky-lating agents, good yields of carboxylic acid esters result. A one-pot procedure is often used, in which the salt is first produced from the carboxylic acid and then directly alkylated to form the ester. Isolation of the salt is necessary only in the case of thermally unstable quaternary ammonium salts
Alkali metal and alkaline-earth metal salts, as well as silver and copper salts, can be used. Dialkyl sulfates, aliphatic halides such as long-chain alkyl chlorides, and alicyclic halides are suitable alkylating agents. The method is used mainly in preparative chemistry.
W. William, B. Halper, Synthesis 1974, 727.
5.3. Acylation with Acyl Halides
High yields of esters are obtained by acylation of alcohols with carboxylic acid halides:

The acyl chloride is often produced in situ. The liberated hydrogen halide is generally bound to a base, e.g., pyridine or alkali hydroxide (Schotten Baumann reaction).
This method is particularly important in preparative and analytical chemistry. A commercial application is the production of polycarbonates from phosgene and diols. Phenyl esters and esters of sterically hindered carboxylic acids can be produced from the corresponding acyl halides in the presence of Lewis acids, e.g., iron(III), aluminum, tin(IV), and titanium(IV) chlorides. Addition of phase-transfer agents such as tetrabutylammonium chloride has also proved effective . An important application of this method is in the esterification of steroids.
G. Schwabe, G. Westphal, H. G. Hennig, Z. Chem. 20 (1980) 184.S. Szeja, Synthesis 1980, 402.
5.4. Acylation with Carboxylic Anhydrides
Carboxylic anhydrides are suitable acylating agents for alcohols and phenols. With the exception of trifluoroacetic anhydride and mixed carboxylic anhydrides, they are less reactive than acyl halides.
The advantage of this method is that no water is formed; however, only one equivalent acyl per molecule of anhydride is available for esterification:

Therefore, the method is especially suitable for the esterification of rare or expensive alcohols with cheap anhydrides such as acetic anhydride.
Acylation can be accelerated by addition of protonic acids (sulfuric acid, hydrochloric acid, perchloric acid, p-toluenesulfonic acid) or Lewis acids (zinc chloride). A large-scale commercial process is the production of cellulose acetate from cellulose and acetic anhydride ( Cellulose Esters - 2.1. Cellulose Acetate
Another important product is acetylsalicylic acid (aspirin), which is obtained from salicylic acid and acetic anhydride.
Base-catalyzed acylation with anhydrides is used to synthesize esters from sterically hindered or acid-sensitive alcohols . Common bases are triethylamine and pyridine.
Reaction with anhydrides is the basis of a quantitative analytical method for hydroxyl groups.
J. Cason, Org. Synth., Coll. Vol. 3 (1955) 169.
W. Steglich, DE 1 958 954, 1969 (W. Steglich, G. Hfle).
5.5. Transesterification
This method has already been described in Section 3.2. Transesterification. Transesterifications take place under extremely mild conditions, which permits the reaction of components containing additional functional groups. Chiral centers can be preserved in many cases.
The most common transesterification catalysts are protonic acids (sulfuric acid, perchloric acid, p-toluenesulfonic acid, and molecular sieves). Alkaline catalysts such as alkoxides or anionic ion exchangers are especially effective in peptide chemistry. Potassium cyanide is recommended for unsaturated esters, which undergo cis trans isomerization in the presence of acids. Thallium(I) salts are also employed. If titani-um(IV) alkoxides or complexes of triphenylphosphine with trialkyltin alkoxides or with copper alkoxides are used, the alkoxide component need not be identical with the alcohol group that is exchanged.
5.6. Acylation with Ketenes
Ketenes react with alcohols or phenols to form carboxylic acid esters:
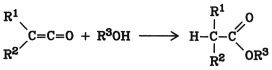
The addition is catalyzed by acids and bases (e.g., pyridine or sodium acetate). The use of ketenes is limited because they are difficult to handle and equally good results can be achieved with other acylating agents such as acyl halides and anhydrides.
The alcoholysis of diketene is an important method for the commercial production of b-oxocarboxylic acid esters (acetoacetates), especially ethyl acetoacetate [141-97-9]
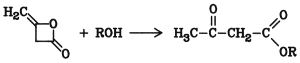
Acetoacetic acid anilides are produced in a similar way. These compounds are used in the synthesis of dyes, especially fast yellow dyes.
Hoechst, DE 1 956 558, 1969 (E. Fischer, O. Mautz).
5.7. Carbonylation
The preparation of carboxylic acid esters by carbonylation of olefins in alcoholic solution (Reppe synthesis) is used on an industrial scale, e.g., in the production of methyl and ethyl propionate ( Carbonylation

The linear ester is obtained in 85 % yield, with less than 10 % branched ester of the following structure:
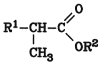
The reaction is carried out at a moderate temperature (ca. 100 C) and a CO pressure of 20 25 MPa (200 250 bar). Iron, nickel, and cobalt salts, which are converted into the corresponding metal carbonyls during the reaction, and palladium or rhodium complexes are used as catalysts. A typical example is a bimetallic complex of bis(triphenylphosphine)palladium(II) chloride and tin(II) chloride . Similarly, 1,3-dienes give g d-unsaturated esters.
Alkyl, benzyl, vinyl, aryl, and allyl halides can also be carbonylated in the presence of suitable catalysts such as Mn Fe Ni powders. These reactions require only a low CO pressure
Carbonylation of alcohols at a pressure up to 70 MPa (700 bar) in the absence of olefins gives formates. Thus, methyl formate is obtained by carbonylation of methanol at 70 C and 2 20 MPa (20 200 bar) in the presence of sodium methoxide
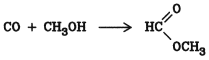
J. Falbe: New Syntheses with Carbon Monoxide, Springer Verlag, Heidelberg 1980.
F. Knifton, J. Org. Chem. 41 (1976) 2885.
C. Cassar, G. P. Chiuroli, F. Guerrieri, Synthesis 1973, 509.
K. Weissermel, H. J. Arpe: Industrielle Organische Chemie, 2nd ed., Verlag Chemie, Weinheim New York 1978, pp. 42 43, pp. 216
5.8. Condensation of Aldehydes
The condensation of two acetaldehyde molecules (Tishchenko reaction) is an important industrial process for the production of ethyl acetate:Aluminum ethoxide promoted with chloride ions serves as a catalyst. In practice, acetaldehyde is passed at 0 5 C through a mixture of aluminum filings and traces of AlCl3 in ethanol ethyl acetate; the yield is 95
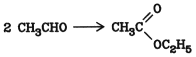
5.9. Alcoholysis of Nitriles
Nitriles react directly with alcohols to produce the corresponding esters:
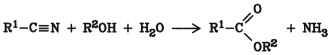
The ammonia that is formed must be removed from the equilibrium, preferably by using an excess of a strong mineral acid, e.g., sulfuric acid or p-toluenesulfonic acid. The acid also acts as a catalyst
This method is used widely in industry for the production of esters from unsaturated nitriles such as acrylonitrile and methacrylonitrile. Alcoholysis of acrylonitrile is carried out with concentrated sulfuric acid. This leads initially to formation of the acrylamide sulfate, which then reacts with the alcohol (preferably methanol or ethanol) to produce the ester:
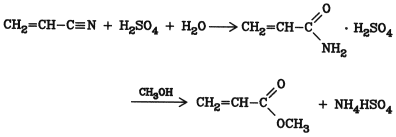
Selectivity for methyl acrylate is > 90 %. A disadvantage of this process is the formation of ammonium hydrogen sulfate, which cannot be used economically. An alternative process, which avoids formation of this byproduct, is the vapor-phase alcoholysis of acrylonitrile in the presence of boric oxide
Methyl methacrylate is obtained by alcohol-ysis of the methacrylonitrile precursor acetone cyanohydrin. In this case, methacrylamide sulfate is first produced by using 98 % H2SO4 at 80 140 C, which is then reacted with methanol at 80 C to produce methyl methacrylate

Selectivity based on acetone is 77 %. Other methods, which avoid the formation of NH4HSO4, have been developed on a pilot-plant scale.
F. L. James, W. H. Bryan, J. Org. Chem. 23 (1958) 1225.
Mitsui Toatsu, JP 4 725 120, 1971.
Rhm & Haas, DE 1 468 939, 1965 (G. Schrder, H. Fink).
5.10. Acylation of Olefins
Direct addition of olefins to carboxylic acids is used on a large scale for the production of vinyl acetate, the ester of the hypothetical vinyl alcohol ( Vinyl Esters
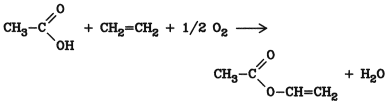
The presence of stoichiometric amounts of oxygen or air is necessary. Palladium or palladium salts are essential as catalysts; other group VIII metals are less effective.
The process can be carried out in the vapor phase or in the liquid phase. Currently, only the vapor-phase process is used on an industrial scale. The reaction conditions are 175 200 C and 0.5 1 MPa (5 10 bar); the catalyst consists of palladium metal and alkali acetate on a carrier. The carrier is necessary for activation and higher selectivity. During the reaction, the alkali acetate is slowly removed from the catalyst and has to be continuously replaced . The vapor-phase process avoids the corrosion problems of the liquid-phase process and gives better yields of vinyl acetate (94 % based on ethylene or 98 % based on acetic acid).
The liquid-phase process is no longer used on an industrial scale. This process is closely related to the Wacker Hoechst acetaldehyde process ( Acetaldehyde - 4.3.1. Direct Oxidation of Ethylene). The catalyst contains copper salts as a redox system. The advantages of the liquid-phase process are a better control of the highly exothermic reaction and the generation of acetaldehyde as a byproduct, which can be oxidized to acetic acid. In this way the necessary acetic acid can be entirely produced from ethylene. The disadvantages are corrosion problems and a lower yield of vinyl acetate, based on ethylene.
The olefin process has replaced the older process based on acetic acid and acetylene, because ethylene is cheaper and more readily available in larger quantities.
Allyl esters are produced from propene, carboxylic acids, and oxygen by using a vapor-phase process. Supported catalysts impregnated with palladium salts and bismuth or cadmium compounds are used . Methallyl acetate is obtained from isobutene and acetic acid in the presence of palladium(II) chloride

Glycol mono- and diesters can be produced from olefins and carboxylic acids by adding stoichiometric amounts of oxygen:

W. Schwerdtel, Chem. Ing. Tech. 40 (1968) 781. Hoechst AG, DE 1 191 366, 1961 (W. Riemenschneider, T. Quadflieg). Hoechst AG, DE-AS 1 618 391, 1967 (L. Hrnig, H. Fernholz, et al.) DE-AS 1 643 999, 1967 (H. Fernholz, H.-J. Schmidt). DE-AS 1 667 140, 1967 (H. Erpenbach, H. Glaser, et al.). Knapsack AG, DE-OS 1 668 352, 1967 (K. Sennewald, W. Vogt, et al.). Asahi Kasei Kogyo, DE 1 768 643, 1968;DE-AS 1 793 362, 1968 (N. Kominami, H. Nakajima). Bayer AG, DE 1 793 519, 1968 (W. Krnig, G. Scharfe). Knapsack AG, DE-AS 1 808 610, 1968 (K. Sennewald, W. Vogt). Hoechst AG, DE-OS 2 315 037, 1973 (H. Fernholz, H. Krekeler, et al.). Celanese Corp., DE-OS 2 361 098, 1973 (G. M. Severs, Jr.). General Electric Co., DE-OS 2 503 748, 1975; 2 503 926, 1975 (W. E. Smith, R. J. Gerhart). H. Krekeler, H. Schmitz, Chem. Ing. Tech. 40 (1968) 785. Hoechst AG, DE-AS 1 668 063, 1967 (G. Roscher, H. Schmitz).
Hoechst AG, DE 1 903 954, 1969 (H. Fernholz, H.-J. Schmidt).Air Products & Chemicals, DE-AS 1 768 770, 1968 (J. G. Schukys).General Electric Co., DE-OS 2 504 230, 1975;2 504 231, 1975 (W. E. Smith, R. J. Gerhart).
ICI, DE 1 468 086, 1965 (D. Clark, D. Wright).
5.11. Ethoxylation of Carboxylic Acids
Addition of ethylene oxide to carboxylic acids generally proceeds smoothly and quickly. The low acidity of the carboxylic acids is sufficient to split the strained three-membered ring; no catalyst is required in the first addition step:

Further ethoxylation yields polyglycol esters; this reaction can be controlled or entirely suppressed by addition of bases such as alkali or amines.

Ethoxylation reactions are widely used in industry. An example is the production of bis(2-hydroxyethyl) terephthalate [6422-86-2] , by controlled ethoxylation of terephthalic acid:
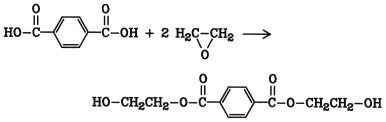
This ester is readily purified by crystallization from water, so that crude terephthalic acid can be used. The ethoxylation is carried out in the liquid phase at 2 3 MPa (20 30 bar) and 90 130 C; no solvent is necessary. The presence of amines or quaternary alkylammonium salts is needed to avoid polyethoxylation. A considerable amount of poly(ethylene terephthalate) is produced by condensation of bis(2-hydroxyethyl) terephthalate.
Polyethoxylation of aliphatic carboxylic acids also is an industrial process. It is carried out under pressure at 120 220 C in the presence of sodium hydroxide or sodium acetate. Addition of 10 30 mol of ethylene oxide per mole of acid causes fatty acids such as stearic acid to lose their hydrophobicity. The products have many applications, e.g., as surfactants. The degree of hydrophilicity depends on the number of glycol units. Ethoxylation of short-chain carboxylic acids yields hydrophilic lubricating oils.
5.12. Orthocarboxylic Acid Esters
Free orthocarboxylic acids are unstable. Their esters can be prepared by reaction of imido ethers with alcohols:
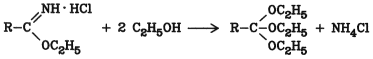
Orthoformic acid ethyl ester [122-51-0] is obtained from chloroform and sodium ethoxide:
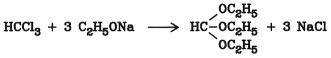
Orthoformates are relatively stable toward alkali; however, they are readily hydrolyzed by acids. In preparative chemistry, orthoformates are used to synthesize acetals and ketals. They also serve for the incorporation of methine groups into compounds with reactive methylene groups.
5.13. Lactones
Lactones are internal esters of hydroxycarboxylic acids. Of commercial interest are b-propiolactone [57-57-8] and g-butyrolactone [96-48-0]. b-Propiolactone can be produced from ketene and formaldehyde in the presence of zinc chloride:
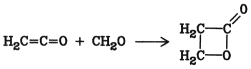
g-Butyrolactone is produced mainly by dehydrogenation of 1,4-butanediol ( Butyrolactone - 4. Production
![]()
Lactones with five- and six-membered rings (g- and d-lactones) are the most stable. They readily form polyesters according to the following equation:

6. Environmental Protection
Most esters are synthesized by esterification of an acid with an alcohol, water being the only byproduct. Special environmental protection measures are usually unnecessary. Most esters possess low toxicity (Chap. 10. Toxicology and Occupational Health). Because many of them are easily hydrolyzed on contact with water or moist air, the toxicological properties of the acid and alcohol component may need to be considered.
Waste Gases. Volatile esters with low carbon numbers are predominantly used as solvents and diluents for lacquers and plastics. Large-scale applications, e.g., in the auto and furniture industries, require excellent ventilation. The solvent vapors are suctioned off and burned in a flare or muffle furnace.
Waste Liquors. Short-chain esters and esters with hydrophilic groups are noticeably soluble in water; some results of biodegradability tests are listed in Table . The toxicity of some common esters for fishes and microorganisms is shown in Table .
7. Quality Specifications and Analysis
Table shows some typical specifications of commercial products.
The most common analytical method is the determination of the saponification value, i.e., the number of milligrams of KOH necessary to hydrolyze 1.0 g of ester:
The ester (0.5 1 g) is refluxed for 30 min with 10 mL of 0.5 N alcoholic KOH. After cooling, the excess alkali is titrated with 0.5 N HCl using phenolphthalein as the indicator.
Before determination of the saponification value, determination of the free acid by titration with alkali in a water alcohol mixture is often necessary.
The ester group can be readily identified by its characteristic absorption bands in the IR spectrum: the strong carbonyl absorption band at ca. 1740 cm1 and two CO stretching bands between 1300 and 1100 cm1
Gas chromatography is suitable only for highly volatile esters. Esters with high boiling points must first be converted into low-boiling esters (usually methyl esters) either by transester-ification or by quantitative hydrolysis and sub-sequent reesterification. Quantitative conversion into the methyl ester can be achieved with diazomethane, methyl iodide calcium oxide in dimethyl sulfoxide (DMSO), or with the boron tri-fluoride methanol complex
Unsaturated esters, e.g., natural fats and oils, are often sufficiently characterized by their iodine value. This is defined as the number of grams of iodine bound by 100 g of an unsaturated ester and is a measure of the number of double bonds in the molecule.
An overview of the different analytical methods is given in . Physical properties such as boiling range, melting point, density, refractive index, residue or ash content, and color are also used to identify esters or mixtures of esters. Previous separation of the different components, (e.g., by high-vacuum distillation) is often unnecessary.
L. J. Bellamy, The Infrared Spectra of Complex Molecules, 3rd ed., J. Wiley & Sons, New York, 1975.L. J. Bellamy, Advances in Infrared Group Frequencies, Methuen & Co., London 1968, pp. 166
L. D. Metcalfe, A. A. Schmitz, Anal. Chem. 33 (1961) 363.
T. S. Ma in S. Patai (ed.): The Chemistry of Carboxylic Acids and Esters, Wiley-Interscience, New York 1969, pp. 871
8. Storage and Transportation
All organic esters are flammable. The flash points, autoignition temperatures, and flammable limits of representative commercial esters are given in Table . The ignition group according to the NFPA (National Fire Prevention Association) corresponds approximately to the danger class of the German VbF (Verordnung ber brennbare Flssigkeiten of 27. Feb.
If esters are stored in tanks, a nitrogen blanket and a carbon filter is necessary. In some cases, the ventilation pipe must be linked to a flare.
Mild steel, aluminum, or other metallic materials can be used for storage and transportation. Plastics are unsuitable because the highly lipophilic esters may migrate into the container walls and cause them to soften or even dissolve. Esters with a high melting point, such as waxes, can also be transported in paper sacks or wooden barrels.
9. Uses and Economic Aspects
Solvents. Because of their highly lipophilic and hydrophobic nature and low polarity, esters are widely used as solvents, extractants, and diluents . Lower esters, especially the acetates of methanol, ethanol, propanols, and butanols, are good solvents for cellulose nitrate and other cellulose derivatives. Ethyl acetate is the most common technical solvent. Branched esters and esters of ethylene glycol also have good solvent properties. Considerable quantities of esters are used as diluents in paints, lacquers, and coatings. Methyl acetate is employed in fast-drying lacquers. Due to the development of low-solvent and solvent-free paints, e.g., for auto paints, the use of esters as diluents is decreasing. Table gives production data for some acetates.
Plasticizers. Large quantities of esters, especially phthalates, adipates, and fatty acid esters, are used as plasticizers . Production data for some important plasticizers are listed in Table . Isooctanoic acid esters and 2-ethylhexanoic acid esters of ethylene glycol are plasticizers for poly(vinyl choride).
Surfactants. Natural fats, oils, and waxes are used in considerable quantities as raw materials in the production of soap (alkali salts of fatty acids) and other detergents and surfactants (Table ). Hydroxyethyl esters of long-chain fatty acids are sold as nonionic surfactants (nonionics), which are easily biodegradable. About 34 100 t of ethoxylated natural fats and oils were produced in the Federal Republic of Germany in 1985, as compared to only 1380 t in Japan. A defined number of ethylene oxide molecules can be added to the fatty acid by varying the process conditions; compounds with a specific water solubility from moderate to excellent can thus be obtained. Nonionics are important as low-foaming detergents and as emulsifiers. They are also used in the food, textile, cosmetics, and pharmaceutical industries. Because of their heat and salt resistance, they are suitable as lubricants in deep-well drilling.
Polyesters. Higher esters derived from natural products, e.g., cellulose acetate and cellulose nitrate, are of considerable economic importance ( Cellulose Esters). However, they are being gradually replaced by synthetic polyesters produced from monomers such as acrylates, te-rephthalates, and vinyl acetate (Table
Vinyl acetate, with a worldwide production of 2106 t/a, is the most important aliphatic ester ( Vinyl Esters). Poly(vinyl acetate) is produced in large quantities for use in plastics, coatings, adhesives, and laminates ( Poly(Vinyl Esters)). A considerable proportion is converted into poly(vinyl alcohol) . Polymerization of acrylates such as methyl acrylate and methyl methacrylate yields homopolymers and mixed polymers used in paints, lacquers, and coatings. Polycondensation of dicarboxylic acids and diols gives polyester resins and plastics. Linear polyesters in particular are produced in large quantities.
Linear polyesters are used in the manufacture of synthetic fibers, packaging films, tapes, films for electric insulation, and a variety of plastics articles such as plastic bottles; they are also employed in hot-melt adhesives.
Polyesters are produced chiefly by transester-ification. The most important raw material is dimethyl terephthalate. This undergoes polycondensation with a diol (e.g., 1,2-ethanediol, 1,2-propanediol, 1,4-butanediol, 1,4-cyclohexanedimethanol), and methanol is released. However, direct esterification of terephthalic acid with ethylene oxide and polymerization of the resulting bis(2-hydroxyethyl) terephthalate is also used (see Section 5.11. Ethoxylation of Carboxylic Acids). Copolymerization with unsaturated acids such as maleic acid yields polymers that can be cross-linked (thermosetting polymers).
Alkyd resins are an important group of polyesters used mainly in lacquers. The stability of the lacquer, e.g., its weatherability, can be improved by adding esters that are highly resistant to hydrolysis, such as pivalic acid esters.
Flavors and Fragrances. Certain esters with a pleasant odor are used in fragrances, flavors, cosmetics, and soaps. Production data for the most important flavor and perfume esters are given in Table . Although the quantities that are used are small, these esters are of great commercial importance because they are expensive . Terpene esters as well as simple esters such as butyl and isobutyl acetate (fruity flavor) and benzyl acetate (jasmine odor) are widely used. These esters can be obtained from plant extracts, but many are currently produced by synthetic processes ( Flavors and Fragrances
Other Uses. Esters can be converted into various derivatives (see Chap. 3. Chemical Properties) and are useful intermediates in preparative chemistry. Ethyl formate is used in the synthesis of vitamin B1. Methyl formate is converted on a large scale into formamide and formic acid; about 30 000 t/a of formic acid are produced by this route.
Natural oils, fats, and waxes are used as lubricants for high-speed engines. High-boiling synthetic esters that experience little change in viscosity when exposed to extremes of temperature, either high or low, have been developed for use in auto and aircraft turboengines. Examples are bis(2-ethylhexyl) sebacate [122-62-3] and glycol or polyglycol esters of branched-chain carboxylic acids. These esters are also used as hydraulic fluids.
An example of the use of esters in pharmaceuticals is the common analgesic aspirin [50-78-2] (2-acetoxybenzoic acid. The active salicylic acid is released only after hydrolysis in the stomach. Benzocaine [94-09-7], the ethyl ester of 4-aminobenzoic acid, is a topical anesthetic. Phenyl salicylate [118-55-8] , Salol, has antipyretic, antirheumatic, and antiparasitic properties.
E. H. Davies, T. H. Durrans: Solvents, 2nd ed., Chapman & Hall, London 1971, pp. 145
C. Marsden, S. Mann (eds.): Solvents Guide, 2nd ed., Interscience Publishers, New York 1963.
D. N. Buttrey: Plasticizers, 2nd American ed., Franklin Publishing Co., Palisades, New York 1960.
I. Mellan: Industrial Plasticizers, Mac Millan, New York 1963.
K. Weissermel, H. J. Arpe: Industrielle Organische Chemie, 2nd ed., Verlag Chemie, Weinheim New York 1978, pp. 42 43, pp. 216
D. L. J. Opdyke: 'Monographs on Fragrance,' Food Cosmet. Toxicol. 16 (1978) Suppl. 1, 839 841; 17 (1979) Suppl., 735 843; 19 (1981) Suppl. 2, 237
10. Toxicology and Occupational Health
With a few exceptions, esters have very low toxicity. Natural fats and oils are widely used in human nutrition. Table lists toxicity data for commercially important esters
The ester group is nontoxic. However, all esters are hydrolyzed more or less rapidly on contact with water; therefore, the toxicity of their hydrolysis products must be taken into account
Most esters readily penetrate skin and mucous membranes. The considerable absorption through the skin can cause health problems. Exposed skin should be thoroughly washed with soap and plenty of water; clothing should be changed.
Inhalation of highly volatile esters in high concentrations can have a narcotic effect. Secondary effects include breathlessness and fits of choking. Ester vapors should not be allowed to contact the eye. Good ventilation of the work area and wearing of breathing apparatus are generally recommended
N. I. Sax: Dangerous Properties of Industrial Materials, 4th ed., Reinhold Publishing Corp., New York 1975.
Threshold Limit Values for Chemical Substances in the Work Environment, ACGIH Cincinnati, 1986.
M. Windholz (ed.): The Merck Index, 10th ed., Merck & Co, Rahway, N.J., 1983.
E. E. Sandmeyer, C. J. Kirwin in G. D. Clayton, F. E. Clayton (eds.): Patty's Industrial Hygiene and Toxicology, 3rd ed., vol. 2 A, Wiley-Interscience, New York 1981, pp. 2259 ff.
11. References
[1] S.Patai(ed.):The Chemistry of Carboxylic Acids and Esters, Wiley-Interscience, New York 1969.J. D'Ans, E. Lax: Taschenbuch fr Chemiker und Physiker, 3rd ed., Springer Verlag, Berlin Heidelberg New York 1970.
[2] J. D. Fletcher, O. C. Dermer, R. B. Fox (eds.): Nomenclature of Organic Compounds, Am. Chem. Soc., Washington D.C. 1974, pp. 137 145.IUPAC commission on nomenclature of organic chemistry: Nomenclature of Organic Chemistry, 4th ed., Pergamon Press, Oxford-New York 1979.
[3] K. S. Markley (ed.): Fatty Acids, Their Chemistry, Properties, Production and Uses, 2nd ed., Interscience, New York 1960/61.
[4] D. Swern (ed.): Bailey's Industrial Oil and Fat Products, Wiley-Interscience, New York 1969, p. 937.
[5] J. Smidt et al., Angew Chem. 74 (1962) 93.
[6] Unilever, DE 2 327 729, 1973 (J. J. Muller, T. J. Kock).
[7] Rohm & Haas, DE 1 932 625, 1969 (W. R. Winslow).
[8] Degussa, DE 1 568 376, 1966 (G. Morlock, H. Trageser).
[9] A. M. Schwartz, J. W. Perry, J. Berch: Surface-Active Agents and Detergents, vol. 2, Interscience, New York 1958, p. 104.
[10] H. Adkins, Org. React. (N.Y.) 8 (1954) 1
[11] N. G. Gaylord: Reduction with Complex Metal Hydrides, New York 1956, pp. 391
[12] C. B. Hauser, B. E. Hudson, Jr., Org. React. (N.Y.) 1 (1947) 266, 302.
[13] J. J. Bloomfield et al., Org. React. (N.Y.) 23 (1976) 259
[14] Houben-Weyl, E5, 656
[15] H. R. Harrison, W. M. Heynes et al., Chem. Ind. (London) 1968, 1568.
[16] J. R. Rachele, J. Org. Chem. 28 (1963) 2898.
[17] I. Roberts, H. C. Urey, J. Am. Chem. Soc. 60 (1938) 2391.
[18] H. Zimmermann, J. Rudolf, Angew. Chem. 77 (1965) 65 74;Angew. Chem. Int. Ed. Engl. 4 (1965) 40.
[19] R. Bader, A. D. Kontowicz, J. Am. Chem. Soc. 75 (1953) 5116.
[20] Knapsack, DE 2 226 829, 1972 (E. Lohmar, F. X. Werber).
[21] G. A. Olah et al., Synthesis 1978, 929 ff.
[22] H. J. Bergk, Z. Chem. 18 (1978) 22 ff.
[23] G. Hallas, J. Chem. Soc. 1965, 5770.
[24] B. F. Goodrich Comp.DE 1 005 947, 1955 (W. L. Bears, F. X. Werber).B. F. Goodrich Comp., DE 1 103 335, 1956 (F. X. Werber, S. J. Averill).Reichhold Chemie AG, DE 1 173 473, 1962 (H. Dalibar).Reichhold Chemie AG, DE-AS 1 185 611, 1963 (H. Dalibar).ICI, DE-AS 1 292 140, 1966 (D. K. Steel).Scholven Chemie AG, DE-OS 1 593 503, 1966 (K. S. Schmitt, W. Flakus et al.).Mobil Oil Comp., DE-AS 1 618 639, 1967 (A. B. Gainor, L. McMakin)British Titan Products Comp., DE-OS 1 807 103, 1968 (R. H. Stanley, D. W. Brook, et al.).Hls AG, DE-AS 2 243 240, 1972 (F. List, K. Wember)F. Brill, L. Ferry, I. T. Baker, High Polym. 27 (1972) 506.
[25] Hls, DE 914 006, 1951 (A. Coenen, F. Broich).
[26] E. Taschner, C. Wasielewski, Liebigs Ann. Chem. 640 (1961) 136
[27] R. C. Parish, C. M. Stock, J. Org. Chem. 30 (1965) 927.
[28] S. Natelson, S. Gottfried, Org. Synth., Coll. Vol. 3 (1955) 381.
[29] Ruhrchemie, DE 1 171 897, 1959 (H. Feichtinger, H. Noerke).
[30] BASF, DE 878 348, 1942 (H. Dierichs, E. Braun, et al.).
[31] W. William, B. Halper, Synthesis 1974, 727.
[32] G. Schwabe, G. Westphal, H. G. Hennig, Z. Chem. 20 (1980) 184.S. Szeja, Synthesis 1980, 402.
[33] J. Cason, Org. Synth., Coll. Vol. 3 (1955) 169.
[34] W. Steglich, DE 1 958 954, 1969 (W. Steglich, G. Hfle).
[35] Hoechst, DE 1 956 558, 1969 (E. Fischer, O. Mautz).
[36] J. Falbe: New Syntheses with Carbon Monoxide, Springer Verlag, Heidelberg 1980.
[37] F. Knifton, J. Org. Chem. 41 (1976) 2885.
[38] C. Cassar, G. P. Chiuroli, F. Guerrieri, Synthesis 1973, 509.
[39] K. Weissermel, H. J. Arpe: Industrielle Organische Chemie, 2nd ed., Verlag Chemie, Weinheim New York 1978, pp. 42 43, pp. 216
[40] F. L. James, W. H. Bryan, J. Org. Chem. 23 (1958) 1225.
[41] Mitsui Toatsu, JP 4 725 120, 1971.
[42] Rhm & Haas, DE 1 468 939, 1965 (G. Schrder, H. Fink).
[43] W. Schwerdtel, Chem. Ing. Tech. 40 (1968) 781. Hoechst AG, DE 1 191 366, 1961 (W. Riemenschneider, T. Quadflieg). Hoechst AG, DE-AS 1 618 391, 1967 (L. Hrnig, H. Fernholz, et al.) DE-AS 1 643 999, 1967 (H. Fernholz, H.-J. Schmidt). DE-AS 1 667 140, 1967 (H. Erpenbach, H. Glaser, et al.). Knapsack AG, DE-OS 1 668 352, 1967 (K. Sennewald, W. Vogt, et al.). Asahi Kasei Kogyo, DE 1 768 643, 1968;DE-AS 1 793 362, 1968 (N. Kominami, H. Nakajima). Bayer AG, DE 1 793 519, 1968 (W. Krnig, G. Scharfe). Knapsack AG, DE-AS 1 808 610, 1968 (K. Sennewald, W. Vogt). Hoechst AG, DE-OS 2 315 037, 1973 (H. Fernholz, H. Krekeler, et al.). Celanese Corp., DE-OS 2 361 098, 1973 (G. M. Severs, Jr.). General Electric Co., DE-OS 2 503 748, 1975; 2 503 926, 1975 (W. E. Smith, R. J. Gerhart). H. Krekeler, H. Schmitz, Chem. Ing. Tech. 40 (1968) 785. Hoechst AG, DE-AS 1 668 063, 1967 (G. Roscher, H. Schmitz).
[44] Hoechst AG, DE 1 903 954, 1969 (H. Fernholz, H.-J. Schmidt).Air Products & Chemicals, DE-AS 1 768 770, 1968 (J. G. Schukys).General Electric Co., DE-OS 2 504 230, 1975;2 504 231, 1975 (W. E. Smith, R. J. Gerhart).
[45] ICI, DE 1 468 086, 1965 (D. Clark, D. Wright).
[46] L. J. Bellamy, The Infrared Spectra of Complex Molecules, 3rd ed., J. Wiley & Sons, New York, 1975.L. J. Bellamy, Advances in Infrared Group Frequencies, Methuen & Co., London 1968, pp. 166
[47] L. D. Metcalfe, A. A. Schmitz, Anal. Chem. 33 (1961) 363.
[48] T. S. Ma in S. Patai (ed.): The Chemistry of Carboxylic Acids and Esters, Wiley-Interscience, New York 1969, pp. 871
[49] E. H. Davies, T. H. Durrans: Solvents, 2nd ed., Chapman & Hall, London 1971, pp. 145
[50] C. Marsden, S. Mann (eds.): Solvents Guide, 2nd ed., Interscience Publishers, New York 1963.
[51] D. N. Buttrey: Plasticizers, 2nd American ed., Franklin Publishing Co., Palisades, New York 1960.
[52] I. Mellan: Industrial Plasticizers, Mac Millan, New York 1963.
[53] D. L. J. Opdyke: 'Monographs on Fragrance,' Food Cosmet. Toxicol. 16 (1978) Suppl. 1, 839 841; 17 (1979) Suppl., 735 843; 19 (1981) Suppl. 2, 237
[54] N. I. Sax: Dangerous Properties of Industrial Materials, 4th ed., Reinhold Publishing Corp., New York 1975.
[55] Threshold Limit Values for Chemical Substances in the Work Environment, ACGIH Cincinnati, 1986.
[56] M. Windholz (ed.): The Merck Index, 10th ed., Merck & Co, Rahway, N.J., 1983.
[57] E. E. Sandmeyer, C. J. Kirwin in G. D. Clayton, F. E. Clayton (eds.): Patty's Industrial Hygiene and Toxicology, 3rd ed., vol. 2 A, Wiley-Interscience, New York 1981, pp. 2259 ff.
[58] D. L. J. Opdyke, Food Cosmet. Toxicol. 12 (1974) 719.
[59] Hoechst AG, unpublished results.
|
Politica de confidentialitate | Termeni si conditii de utilizare |

Vizualizari: 6142
Importanta: ![]()
Termeni si conditii de utilizare | Contact
© SCRIGROUP 2025 . All rights reserved