OXYGEN
& CARBON DIOXIDE TRANSPORT BY BLOOD
General Goal To
describe the storage of oxygen and carbon dioxide in blood and tissues, and to
provide a framework for understanding how these stores interact.
Specific Objectives The student should:
be able to name the major regions of  drop as blood moves from alveolus to
mitochondria.
drop as blood moves from alveolus to
mitochondria.
know the shape of the myoglobin dissociation
curve, and be able to state the two major functions of myoglobin.
know what is meant by the P50 of
hemoglobin, be able to sketch a normal oxygen dissociation curve, and be able
to state the significance of the flat portion for oxygen loading, and the steep
portion for oxygen unloading.
be able to define oxygen carrying capacity,
oxygen content, oxygen saturation, and oxygen partial pressure.
be able to describe the effects of pH, 2,3-DPG,
temperature 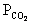 ,
and
,
and 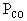 on
hemoglobin P50.
on
hemoglobin P50.
be able to describe the effects of
methemoglobinemia, carboxyhemoglobin, and anemia on oxygen carrying capacity.
be able to define the Bohr effect, the Haldane
effect, and the chloride shift.
be able to quantitate the way in which CO2
is carried in plasma and the red cells.
be able to describe the significant differences
between the oxygen dissociation curve and the carbon dioxide dissociation
curve.
Resources Lecture: Dr. Baer
Reading: West,
JB. Respiratory Physiology-The
Essentials (4th Ed.). Chapter
6.
Mines, AH. Respiratory Physiology. Raven Press. Chapters 4, 5, & 6.
I.
OXYGEN TENSIONS FROM
ALVEOLI TO MITOCHONDRIA
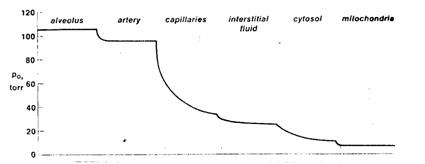
Figure 1. The transport of oxygen in higher organisms
requires the presence of concentration gradients at several sites, with the
oxygen tension progressively falling from the lung alveoli to the mitochondria
in the peripheral tissues where oxygen is consumed.
A.
Although oxygen tension falls in several places,
the greatest fall in oxygen tension occurs across the systemic capillaries.
B.
Mixed venous 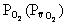 is
normally 40 mmHg.
is
normally 40 mmHg.
Some tissues extract a greater proportion of
oxygen from arterial blood and have a 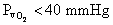 .
.
Some tissues extract a lesser proportion of the
oxygen from arterial blood and have a 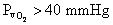 .
.
C.
The partial pressure of oxygen in the
mitochondria is often considerably lower than the  at the end of the systemic capillary. It is therefore unfair to use the
at the end of the systemic capillary. It is therefore unfair to use the 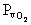 of an organ as a measure of its cellular
of an organ as a measure of its cellular  .
.
D.
Diffusion-limited
transport. The existence of an end-capillary gradient  in some peripheral tissues is indicative of
diffusion limited transport.
in some peripheral tissues is indicative of
diffusion limited transport.
E.
The total body oxygen stores include:
oxygen in the cells.
oxygen in the blood.
oxygen in the lung.
II.
CELLULAR OXYGEN STORES
AND MYOGLOBIN
A.
Cells without myoglobin have oxygen stores
limited by oxygens solubility.
B.
Myoglobin is a protein which binds oxygen in red
muscle. Red muscle includes the heart
which is a continuously working aerobic organ and red skeletal muscle used in
the performance of aerobic or endurance exercise.
C.
Myoglobin Functions. There are 2 probable functions.
Buffer changes in tissue  during changes in cellular metabolism.
during changes in cellular metabolism.
Facilitate O2 diffusion through the
cytoplasm to the mitochondria where the O2 is used.
D.
Myoglobin dissociation curve.
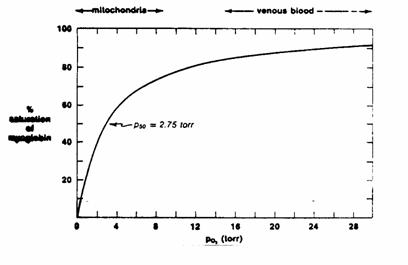
Figure 2
Myoglobin is a single polypeptide chain
containing 1 heme group. Each molecule
of myoglobin binds one molecule of O2.
Myoglobin binding kinetics obey simple mass
action laws and are therefore Michaelis-Menton kinetics.
The P50 of myoglobin is the partial
pressure at which 50% of the myoglobin molecules (or 50% of the myoglobin
binding sites) have bound O2. This is by analogy to the Km of an enzyme.
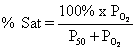
At venous  levels, myoglobin is nearly fully saturated.
levels, myoglobin is nearly fully saturated.
At mitochondrial  levels, a small fall in
levels, a small fall in  causes myoglobin to release most of its oxygen
for use (steep part of curve).
causes myoglobin to release most of its oxygen
for use (steep part of curve).
E.
Myoglobin is also thought to facilitate
intracellular O2 transport.
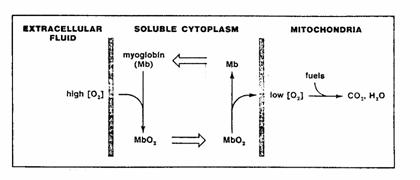
Figure 3. The presence of myoglobin can facilitate
diffusion within a cell. If the
concentration gradient of O2 is in a range over which the
concentration of oxygen greatly affects the degree of its binding to myoglobin,
much more of the myoglobin will be oxygenated on the high side of the
gradient. Effective concentration
gradients will therefore be established for oxymyoglobin (MbO2) in
one direction and for deoxymyoglobin (Mb) in the opposite direction, so that
molecules of the carrier will go to and fro across the concentration gradient,
picking up O2 on the high side, and releasing it on the low.
III.
BLOOD OXYGEN STORES AND
HEMOGLOBIN
A.
Oxygen is carried in blood in 2 forms
Dissolved in plasma. Normally insignificant.
Bound to hemoglobin in red blood cells.
B.
Ways of expressing the amount of oxygen in
blood.
O2
Partial Pressure. (Also called oxygen
tension.) Units are mmHg. The partial pressure of oxygen is
proportional to the amount of O2 dissolved in blood. Partial pressure differences are of
significance in determining the rate of diffusion between blood and tissue.
O2
Content. Units are ml O2/dl blood. These are often written ml/dl. An older synonym is Vol%. By convention volume units are substituted
for molar units (moles/liter) although the later could be calculated by the gas
law. O2 content is the total
amount of oxygen in a deciliter of blood and includes both dissolved and
hemoglobin-bound oxygen, but dissolved [Hb] is normally negligable. Ignoring dissolved Hb:
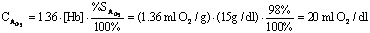
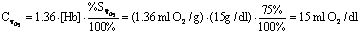
O2
Saturation. This is the fraction or percentage of all the
hemoglobin binding sites that are currently occupied by oxygen. Units are usually percent.
Since
dissolved O2 content is normally negligible, saturation can be
thought of as content/capacity.
C.
Dissolved O2. The arterial plasma normally contains:
D.
Hemoglobin contains 2 alpha chains and 2 beta
chains each of which contains 1 heme group. One molecule of hemoglobin can bind 4 molecules of O2.
One g of hemoglobin can bind 1.36 ml O2. (The numbers 1.34 ml O2 and 1.39
ml O2 are also in common use.)
Blood normally contains 15 g/dl of hemoglobin.
From above, the oxygen binding capacity of blood
with a normal hemoglobin is:
E.
When breathing 100% O2  dissolved O2 contributes 1.8 ml O2/dl
blood.
dissolved O2 contributes 1.8 ml O2/dl
blood.
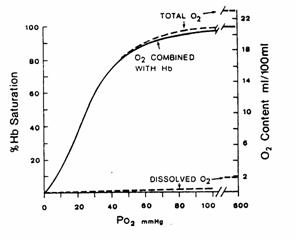
Figure 4. O2 dissociation curve (solid line)
for pH 7.4 PCO2 40 mmHg and
37C. The total blood O2
content is also shown for a hemoglobin concentration of 15 g/100 ml of blood.
F.
Sigmoid binding
curve. Cooperativity causes the oxygen dissociation
curve to display sigmoid kinetics.
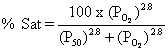
The Hill coefficient is 2.8 (not the expected 4)
because cooperativity is not complete.
The P50 for hemoglobin (half the
binding sites saturated) is 27 mmHg as opposed to 2.8 mmHg for myoglobin.
If there were no cooperativity (Hill coefficient
of 1) and a P50 = 27 mmHg, then hemoglobin would take up less O2
at the lungs (only 79% saturated at  )
and would give up less O2 as
)
and would give up less O2 as  fell to venous levels.
fell to venous levels.
G.
Significance of the Sigmoid curve
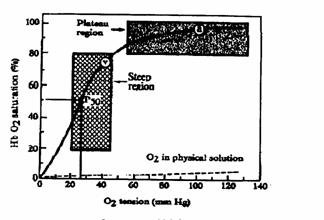
Figure 5. Oxygen-hemoglobin dissociation curve. The driving pressure for loading O2 onto Hb is arterial O2
tension, which is represented by the amount of O2 physically
dissolved in solution: a = arterial; v = venous; P50 = O2
tension required to saturate 50% of the hemoglobin.
Hemoglobin is 90% saturated even when arterial  falls to 60 mmHg. This is because the upper portion of the
curve is relatively flat.
falls to 60 mmHg. This is because the upper portion of the
curve is relatively flat.
In systemic capillaries where blood  may be low
may be low 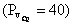 hemoglobin becomes desaturated, i.e., it
releases oxygen for uptake by cells. This is the steep portion of the curve.
hemoglobin becomes desaturated, i.e., it
releases oxygen for uptake by cells. This is the steep portion of the curve.
Normally hemoglobin gives up less than half its
O2. There is reserve in the
system. If systemic capillary  falls further (eg. in exercise) there is
plenty of additional O2 available from hemoglobin.
falls further (eg. in exercise) there is
plenty of additional O2 available from hemoglobin.
H.
Tabular oxygen dissociation curve (*remember
these to sketch your own)
*Values needed reasonably accurate
dissociation curve. Remember that the
PO2 at which the saturation is 50% (the P50, detailed later) 27
mmHg.
|
|
Blood 
|
%O2 Saturation
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
IV.
CHANGES IN HEMOGLOBIN
AFFINITY FOR OXYGEN
A.
Changes in hemoglobin affinity for oxygen result
in a change in P50. Note that
the maximum oxygen capacity is not changed. (change in Km not Vmax.)
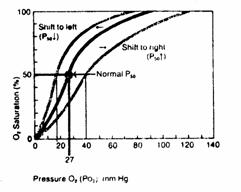
Figure 6
Increased
hemoglobin affinity for O2. The oxygen dissociation curve shifts to the left. P50 is reduced.
Decreased
hemoglobin affinity for O2. The oxygen dissociation curve shifts to the right. P50 is increased.
B.
Shifts in oxyhemoglobin dissociation curve have
minimal effects on oxygen loading in the lung because the oxygen dissociation
curve is still fairly flat at a  of 100
mmHg.
of 100
mmHg.
C.
Shifts in the oxyhemoglobin dissociation curve
have a maximal effect on oxygen unloading at the tissues because the curve is
steep at venous  (eg. 40
mmHg).
(eg. 40
mmHg).
D.
Hydrogen Ion. Increased [H+] (decreased pH)
increases the P50 (decreased O2 affinity) and shifts the
dissociation curve to the right. Decreasing
[H+] the opposite effect.
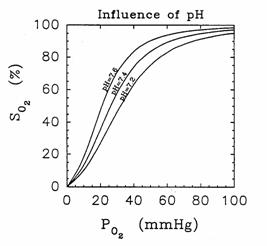 Figure 7
Figure 7
Acidosis promotes oxygen
unloading. At 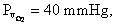 compare
compare 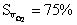 at pH = 7.4 with
at pH = 7.4 with 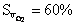 at pH = 7.2.
at pH = 7.2.
Alkalosis inhibits oxygen unloading.
E.
Carbon dioxide (Bohr Effect). Increasing 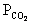 increases P . Decreasing
increases P . Decreasing 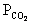 decreases P
decreases P
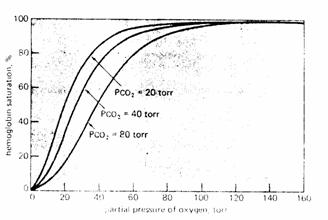
Figure 8
The major portion of the
Bohr Effect is due to the fact that increasing 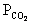 causes a decreased red cell pH (acidosis).
causes a decreased red cell pH (acidosis).
A secondary part of the Bohr Effect is due to
the fact that CO2 reacts covalently with hemoglobin to form
carbamino hemoglobin which has a reduced O2 affinity.
The Bohr Effect plays a significant role in
oxygen unloading under normal, physiological conditions. The P50 of a venous oxygen
dissociation curve may be as high as 35 mmHg as compared to the normal arterial
value of 27 mmHg.
F.
Temperature. Increasing temperature increases P50. Decreasing temperature decreases P50.
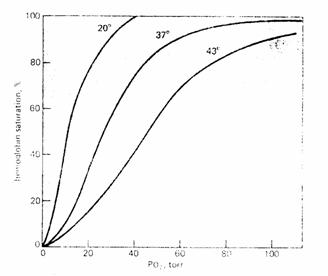
Figure 9
G.
Exercise. Increase temperature, increased 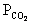 and decreased pH all promote oxygen unloading at
the tissues by increasing P50 (decreased affinity).
and decreased pH all promote oxygen unloading at
the tissues by increasing P50 (decreased affinity).
H.
2,3-diphosphoglycerate
(2,3-DPG). 2,3-DPG is a glycolytic intermediate which
accumulates to uniquely high levels in red cells. Increased levels increase P50;
decreased levels decrease P50. Increased levels occur in association with hypoxia.
Increased levels of 2,3-DPG may be seen in:
a)
acclimatization to high altitudes.
b)
chronic lung disease; emphysema.
c)
anemia.
d)
hyperthyroidism.
e)
right to left cardiac shunt.
f)
congenital heart disease.
g)
pulmonary vascular disease.
Blood bank
storage. Blood storage in citrate-phosphate-dextrose
solution for as short as 1 week can lead to significant 2,3-DPG depletion and
left-shifted oxygen dissociation curves.
Fetal
hemoglobin. 2,3-DPG levels do not affect the P50
of fetal hemoglobin.
V.
CHANGES IN O2
CARRYING CAPACITY OF HEMOGLOBIN
A.
Hemoglobin Concentration. Hemoglobin concentration will change from its
normal value of 15 g/dl as hematocrit changes.
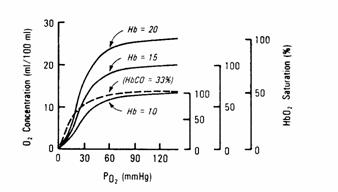
Figure 10
Anemia will decrease
the oxygen carrying capacity of blood without independently altering the P50
of blood.
Polycythemia will increase
the oxygen carrying capacity of blood without independently altering the P50
of blood.
B.
Carbon
Monoxide. Carbon monoxide binds hemoglobin at the
oxygen binding sites to form carboxyhemoglobin. It has approximately 210 times the affinity of oxygen.
Figure 11.Dissociation
curves for HbO2 and HbCO. Dissociation curves are plotted on the same scale. Maximal saturation of hemoglobin with O2
is not reached until the PO2 is greater than 120 mmHg; with CO,
however, maximal saturation is attained with PCO of less than 1
mmHg.
Occupation of binding sites by CO effectively
eliminates them for O2 binding and therefore reduces O2
binding capacity (see figure in Section A above).
Because of the complex cooperativity between
hemoglobin subunits, binding of some sites by CO also reduces P50
for oxygen binding. Note that this does
not occur with anemia.
C.
Methemoglobin. The heme groups of hemoglobin normally
contain ferrous iron (Fe++) whether bound to oxygen or not.
Oxidizing
Agents. Certain drugs and chemicals (eg. nitrates,
acetanilide, sulfonamides) can oxidize the heme Fe++ to Fe+++. The resulting hemoglobin is called
methemoglobin and does not bind O2. The total oxygen carrying capacity is reduced by the amount of
hemoglobin that is methemoglobin.
A certain amount of methemoglobin forms
spontaneously. This is normally less
than 1% of total hemoglobin. The enzyme
NADH-methemoglobin reductase helps prevent accumulation of methemoglobin.
VI.
CARBON DIOXIDE STORES
A.
The solubility of carbon dioxide is 0.075 ml CO2/dl/mmHg.
B.
Carbon dioxide is carried in the plasma in two
forms.
Dissolved CO2. At  the dissolved CO2 concentration is
3.4 ml/dl.
the dissolved CO2 concentration is
3.4 ml/dl.
Carbamino
compounds. Plasma protein concentration is about
7%. CO2 binds the amine
groups of plasma proteins to form carbamino compounds. The hydrogen ions formed are buffered by
plasma proteins.

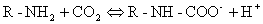
Plasma has little carbonic anhydrase so CO2
forms little carbonic acid in plasma.
C.
Carbon dioxide is carried by the red blood cell
in three forms.
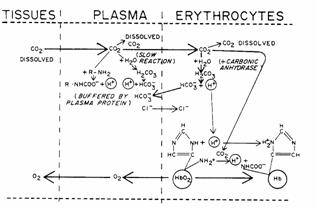
Figure 12
Dissolved CO2. CO2 can cross the red cell
membrane and dissolve in RBC water.
Carbamino
compounds. Approximately 30% of RBC contents is
hemoglobin. CO2 can form
carbamino hemoglobin on amine groups. The H+ released by this reaction is buffered by histidine
residues (imidazole group) on the hemoglobin itself.
Bicarbonate. Carbonic anhydrase is present in RBCs and
catalyze the formation of carbonic acid which dissociated to hydrogen ion and
bicarbonate. The H+ is
buffered by hemoglobin.
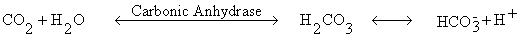
D.
Chloride Shift. As 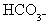 is formed
it diffuses out of the red cell. Cl-
diffuses into the red cell to maintain electroneutrality. This is the Chloride Shift or Hamburger
Shift.
is formed
it diffuses out of the red cell. Cl-
diffuses into the red cell to maintain electroneutrality. This is the Chloride Shift or Hamburger
Shift.
The chloride shift is rapid and is complete
before the cells exit the capillary.
The osmotic effect of the extra 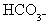 and Cl-
in venous red cells causes the venous RBC volume to increase slightly. For this reason venous hematocrit slightly
exceeds arterial hematocrit.
and Cl-
in venous red cells causes the venous RBC volume to increase slightly. For this reason venous hematocrit slightly
exceeds arterial hematocrit.
E.
Quantitative summary of CO2
transport.
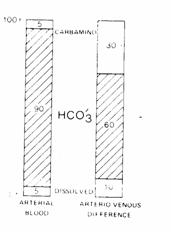
Figure 13
90% of arterial CO2
stores are carried as 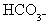 with 5% of the stores carried as
dissolved CO2, and 5% of the stores as carbamino compounds.
with 5% of the stores carried as
dissolved CO2, and 5% of the stores as carbamino compounds.
Of the CO2
added in systemic capillaries, 60% is added as 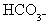 , 30% is added as carbamino compounds and
10% is added as dissolved CO2.
, 30% is added as carbamino compounds and
10% is added as dissolved CO2.
VII.
CARBON DIOXIDE
DISSOCIATION CURVE
A.
The total CO2 content of blood can be
plotted as a function of 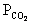
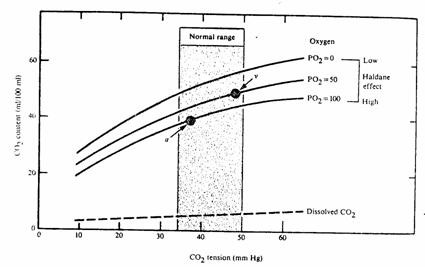
Figure 14
B.
Haldane effect. Increasing O2 tension decreases
the affinity of hemoglobin for CO2. As a result the CO2 dissociation curve shifts downward.
High  promotes CO2 unloading in the
lungs.
promotes CO2 unloading in the
lungs.
Low  promotes CO2 loading in the
periphery.
promotes CO2 loading in the
periphery.
C.
Comparison of CO2 and O2
dissociation curves.
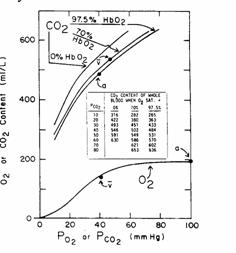
Figure 15
CO2 content is far higher than O2
content at physiological partial pressures.
The CO2 dissociation curve is
approximately linear (a straight line) in the range of arterial and venous
blood. Thus, if equal volumes of blood
of 2 different CO2 contents are mixed together, the resulting 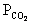 will be
half way between the starting
will be
half way between the starting 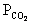 values.
values.
The O2 dissociation is curved between
arterial and venous points. Thus, if
equal volumes of blood of 2 different O2 contents are mixed
together, the resulting blood will have a  which is
not centered between the original
which is
not centered between the original  values.
values.
VIII.
CARBON DIOXIDE AND
ACID-BASE BALANCE
A.
The lungs excrete 13,000 mEq a day of CO2
representing an equivalent volume of carbonic acid. The kidneys excrete 40-80 mEq/day fixed acid.
B.
Henderson-Hasselbach Equation
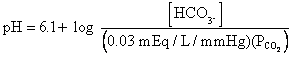
C.
Changes in 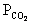 cause changes in [H+] by mass action.
cause changes in [H+] by mass action.
An increase in 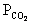 results in respiratory acidosis.
results in respiratory acidosis.
An decrease in 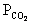 results in respiratory alkalosis.
results in respiratory alkalosis.
D.
The effect of altered 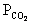 on pH depends on whether the bicarbonate buffer
system acts alone or in concert with other buffer systems.
on pH depends on whether the bicarbonate buffer
system acts alone or in concert with other buffer systems.
TABLE 1. Effects
of adding CO2 to fluids with and without buffers other than
bicarbonate
|
Type of Fluid
|
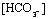
(mEq/liter)
|
pH
|
[H+]
(nEq/liter)
|
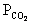
(mmHg)
|
|
|
A. Fluid with no nonbicarbonate
buffers of importance (CSF)
|
|
|
|
|
INITIAL
FINAL
|
|
B. Fluid with buffers in
addition to bicarbonate (blood)
|
|
|
|
|
INITIAL
FINAL
|
CSF. The bicarbonate
buffer system works alone and 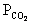 has a larger effect on pH.
has a larger effect on pH.
Blood. Hemoglobin buffers H+ changes in addition to 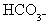 Thus,
changes in
Thus,
changes in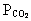 have somewhat blunted effect on pH.
have somewhat blunted effect on pH.
STUDY QUESTIONS FOR OXYGEN AND CARBON DIOXIDE
TRANSPORT
What is meant by the term P50?
What is the Bohr Effect? The Haldane Effect?
List four factors which will increase the P50
for hemoglobin. What is the functional
significance of an increase P50 (rightward shift in the O2
dissociation curve)?
How will each of the following affect 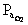 and oxygen content of arterial blood
and oxygen content of arterial blood 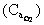 ? a)
Anemia; b) Polycythemia; c) Low
? a)
Anemia; b) Polycythemia; c) Low  in inspired air; d) 2,3 DPG; e) Increased
in inspired air; d) 2,3 DPG; e) Increased 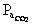 ;
f) Fever; g) Carbon monoxide
poisoning; h) Methemoglobinemia
;
f) Fever; g) Carbon monoxide
poisoning; h) Methemoglobinemia
What effect does oxygen uptake in the lungs have
on the buffering capacity of blood? What
does oxygen unloading in tissues have?
How does oxygen unloading in tissues affect
carbon dioxide uptake by blood?
How does oxygen loading in lungs affect carbon
dioxide release from blood?
What role does the chloride shift play in carbon
dioxide transport by blood?
In what form is carbon dioxide carried in
blood? What is the percent contribution
of each?
Calculate the oxygen carrying capacity of a
person with hemoglobin concentration of 18 g/dl. What is his approximate arterial saturation
if his 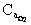 is 17
ml/dl?
is 17
ml/dl?
In a normal person, what would be the O2 saturation at a  of 40 mmHg? At 27 mmHg? At 60 mmHg?
of 40 mmHg? At 27 mmHg? At 60 mmHg?
Of what
significance is the fact that the CO2 dissociation curve is linear
over the normal range? Hint: how does this affect mixing of blood?
![]() drop as blood moves from alveolus to
mitochondria.
drop as blood moves from alveolus to
mitochondria.![]() ,
and
,
and ![]() on
hemoglobin P50.
on
hemoglobin P50.

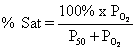

![]()
![]()
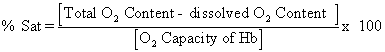
![]()
![]()

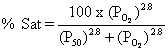


 Figure 7
Figure 7
![]()



![]()
![]()

![]()



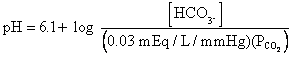
![]() and oxygen content of arterial blood
and oxygen content of arterial blood ![]() ? a)
Anemia; b) Polycythemia; c) Low
? a)
Anemia; b) Polycythemia; c) Low ![]() in inspired air; d) 2,3 DPG; e) Increased
in inspired air; d) 2,3 DPG; e) Increased ![]() ;
f) Fever; g) Carbon monoxide
poisoning; h) Methemoglobinemia
;
f) Fever; g) Carbon monoxide
poisoning; h) Methemoglobinemia![]() is 17
ml/dl?
is 17
ml/dl?![]() of 40 mmHg? At 27 mmHg? At 60 mmHg?
of 40 mmHg? At 27 mmHg? At 60 mmHg?
![]()