| CATEGORII DOCUMENTE |
| Bulgara | Ceha slovaca | Croata | Engleza | Estona | Finlandeza | Franceza |
| Germana | Italiana | Letona | Lituaniana | Maghiara | Olandeza | Poloneza |
| Sarba | Slovena | Spaniola | Suedeza | Turca | Ucraineana |
DOCUMENTE SIMILARE |
||||||
|
||||||
TERMENI importanti pentru acest document |
||||||
| : | ||||||
Environmental and Community Biology
Fern
Sporebank at the Royal Botanic Gardens
Introduction
Fern dust was first investigated scientifically by Marcello Malpighi (1628 1694) who investigated microscopically the dark spots on the underside of fern leaves (Moran, 2004). The complete life cycle of the fern was not discovered until 1848 when Leszczyc-Suminski first described the archegonia as a female sexual organ and from this the life cycle of ferns as is it is today was deduced (Wagenitz, 1999). Ferns belong to the vascular plant group and possess true leaves but lack flowers and seeds. They possess free living 1n gametophytes which produce gametes by mitosis, often both sperm and egg on the same prothallus. A fertilised egg grows to become a 2n sporophyte, the stage universally recognised as being the fern. This produces spores in sporangia on the leaf underside (fig 1 + 2). There are two distinct types of spores, green and non-green. The green spores contain chlorophyll and are ready to germinate and grow as soon as they hit the ground. Unfortunately these spores are often short lived and lose viability within a few weeks although viability of up to a year has been recorded. Non-brown spores usually remain viable for a few months to several decades (Lloyd et al, 1970)
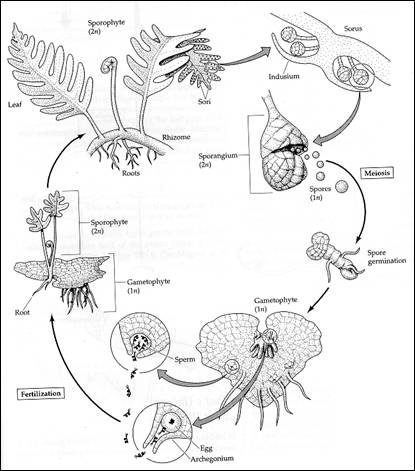
Fig. 1: The Lifecycle of Ferns (www.esu.edu)
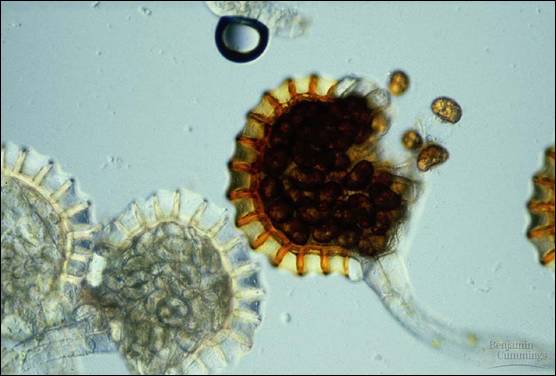
Fig. 2: mature sporangium releasing spores (io.uwinnipeg.ca)
Ferns are not as valuable economically as seed bearing plants but some have historic medical values, Dryopteris filix-mas was once used as an anthelmintic to expel worms and is referred to in Nordic literature (Waller et al, 2001), some have been used as food such as Matteuccia struthiopteris, and some as an aid to nitrogen fixation but most are purely of horticultural interest as specimen and pot plants. Some ferns are now recognized as keystone species and support large varieties of invertebrate life (Ellwood et al, 2002)
Today 193 species of true fern (Pteridophyta) are listed on the IUCN Red List of Threatened Species (IUCN, 2007). As an aid to conversation it must be noted that spores can survive for significant amounts of time in the soil beneath a fern colony, only germinating when exposed to light, this is known as the soil spore bank and has implications in restoring lost species (Dyer, 1994). In order to ensure conservation of certain species, various organisations, usually botanical gardens now operate spore banks collecting and storing spores for use in the future similar to the seed banks held in the same institutions. Methods of storage vary greatly and affect the viability of the spores. Some spores have been known to germinate after 70yrs (Lloyd et al, 1970). The common variables among storage are moisture and temperature. Dry storage involves storing spores in glass vials, wet storage on agar plates and the temperature ranges from +20oC to -80oC. All these variables have effects on the viability of the spores (Quintanilla et al, 2002).
This study will look at
the viability of spore samples subjected to differing storage methods taken
from the Royal Botanic Gardens,
Spore Samples
Dry and cold stored at -20oC. These spores have been less than ideally stored, often with extreme temperature fluctuations (unnoticed freezer breakages and transportation between stores).

Dryopteris affinis a southern and western European native: two samples, one from 1970 and one from 1990.
 Athyrium filix-femina a temperate Northern Hemisphere native: sample
collected 1986
Athyrium filix-femina a temperate Northern Hemisphere native: sample
collected 1986

Dryopteris filix-mas one of the most common ferns occurring in the Northern Hemisphere: sample collected in 1987
Cold Store at 4oC These spores have been stored in spore envelopes and refrigerated

Woodsia ilvensis a small fern found on rocky outcrops above 350m. Currently
there are less than 100 plants remaining in
Fresh Spores Stored Refrigerated

Todea barbara tropical rain forest fern native to Africa,
Method
Notes on Method
I am unsure as to what medium to cultivate the spores on but a simple mix is described in The American Fern Journal: my laboratory has been successfully using a medium consisting of a solution of 15g of agar per litre of water in which is dissolved one commercial nutrient tablet. The resulting medium provides mineral nutrient yielding fern prothallia in numbers and size equal to those grown on conventional nutrient media requiring precise quantitative mineral additives.(Marengo, 1979)
It is also interesting to note that red light stimulates spore growth in the majority of fern species (Kiss, 1998) perhaps the germination rate would be increased if the plates were kept under a red light as opposed to natural light.
References
Dyer A., 1994, Natural soil spore banks - can they be used to
retrieve lost ferns? Biodiversity and Conservation 3,
Ellwood
M., Jones D., Foster W., 2002, Canopy Ferns in
Biotropica Association for Tropical Biology
IUCN 2007. 2007 IUCN Red List of Threatened Species. <www.iucnredlist.org>. Downloaded on 08 February 2008.
Kiss H; Kiss J. 1998
Spore Germination in Populations of Schizaea pusilla from New Jersey and Nova Scotia International Journal of Plant Sciences, Vol. 159, No. 5.
available at: https://links.jstor.org/sici?sici=1058-5893%28199809%29159%3A5%3C848%3ASGIPOS%3E2.0.CO%3B2-B
accessed 08/02/08
Lloyd R; Edward J. Klekowski, Jr. 1970, Spore Germination and Viability in Pteridophyta: Evolutionary Significance of Chlorophyllous Spores, Biotropica, Vol. 2, No. 2.
available at: https://links.jstor.org/sici?sici=0006-3606%28197012%292%3A2%3C129%3ASGAVIP%3E2.0.CO%3B2-Q
accessed 08/02/08
Marengo N. 1979, A Simplified Nutrient Medium for Growing Fern Prothallia American Fern Journal Vol. 69, No. 4
Available at: https://links.jstor.org/sici?sici=0002-8444%28197910%2F12%2969%3A4%3C122%3AASNMFG%3E2.0.CO%3B2-Q
Accessed 07/02/08
Moran R. 2002, A Natural History of Ferns,
Quintanilla L., Amigo J., Panguae. , Pajaron S. 2002 Effect of Storage Method on Spore Viability in Five Globally Threatened Fern Species, Annals of Botany 90 V.4
Available at: https://aob.oxfordjournals.org/cgi/content/full/90/4/461
Accessed 09/02/08
UK Biodiversity Group, 1998, UK Biodiversity Group Tranche 2 Action Plans - Volume I: Vertebrates and vascular plants, Species Action Plan Oblong Woodsia (Woodsia ilvensis). Available online at https://www.ukbap.org.uk/GenPageText.aspx?id=52
Accessed 09/02/08
Wagenitz
G. 1999, Botanical Terminology and
homology in their
Waller P.,Bernes G,Thamsborg SM,Sukura A.,Richter SH,Ingebrigtsen K,Hglund J., 2000, Plants as De-Worming Agents of Livestock in the Nordic Countries: Historical Perspective, Popular Beliefs and Prospects for the Future, published online 2001
Available at: www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2202332
accessed: 08/01/08
|
Politica de confidentialitate | Termeni si conditii de utilizare |

Vizualizari: 4619
Importanta: ![]()
Termeni si conditii de utilizare | Contact
© SCRIGROUP 2026 . All rights reserved