| CATEGORII DOCUMENTE |
| Bulgara | Ceha slovaca | Croata | Engleza | Estona | Finlandeza | Franceza |
| Germana | Italiana | Letona | Lituaniana | Maghiara | Olandeza | Poloneza |
| Sarba | Slovena | Spaniola | Suedeza | Turca | Ucraineana |
Basic Properties of Petroleum
This Chapter describes the physical and chemical properties, which have the greatest bearing on the hazards arising from handling petroleum liquids. These properties are vapor pressure, the flammability of the gases evolved from the liquids and the density of these gases.
VAPOR PRESSURE
True Vapor Pressure
All crude oils and the usual petroleum products are essentially mixtures of a wide range of hydrocarbon compounds (i.e. chemical compounds of hydrogen and carbon). The boiling points of these compounds range from - 162 C (methane) to well in excess of +400 C, and the volatility of any particular mixture of compounds depends primarily on the quantities of the more volatile constituents (i.e. those with a lower boiling point).
The volatility (i.e. the tendency of a crude oil or petroleum product to produce gas) is characterized by the vapor pressure. When a petroleum mixture is transferred to a gas free tank or container it commences to vaporize, that is, it liberates gas into the space above it. There is also a tendency for this gas to re-dissolve in the liquid, and an equilibrium is ultimately reached with a certain amount of gas evenly distributed throughout the space. The pressure exerted by this gas is called the equilibrium vapor pressure of the liquid, usually referred to simply as the vapor pressure.
The vapor pressure of a pure compound depends only upon its temperature. The vapor pressure of a mixture depends on its temperature, constituents and the volume of the gas space in which vaporization occurs; that is, it depends upon the ratio of gas to liquid by volume.
The True Vapor Pressure (TVP) or bubble point vapor pressure is the equilibrium vapor pressure of a mixture when the gas/liquid ratio is effectively zeroed. It is the highest vapor pressure, which is possible at any specified temperature.
As the temperature of a petroleum mixture increases its TVP also increases. If the TVP exceeds atmospheric pressure the liquid commences to boil.
The TVP of a petroleum mixture provides a good indication of its ability to give rise to gas. Unfortunately it is a property which is extremely difficult to measure, although it can be calculated from a detailed knowledge of the composition of the liquid. For crude oils it can also be estimated from the stabilization conditions, making allowance for any subsequent changes of temperature or composition. In the case of products, reliable correlations exist for deriving TVP from the more readily measured Reid Vapor Pressure and temperature.
Reid Vapor Pressure
The Reid Vapor Pressure (RVP) test is a simple and generally used method for measuring the volatility of petroleum liquids. It is conducted in a standard apparatus and in a closely defined way. A sample of the liquid is introduced into the test container at atmospheric pressure so that the volume of the liquid is one fifth of the total internal volume of the container. The container is sealed and immersed in a water bath where it is heated to 37.8 C. After the container has been shaken to bring about equilibrium conditions rapidly, the rise in pressure due to vaporization is read on an attached pressure gauge. This pressure gauge reading gives a close approximation, in bars, to the vapor pressure of the liquid at 37.8 C.
RVP is useful for comparing the volatility of a wide range of petroleum liquids in a general way. It is, however, of little value in itself as a means of estimating the likely gas evolution in specific situations, mainly because the measurement is made at the standard temperature of 37.8 C and at a fixed gas/liquid ratio. For this purpose TVP is much more useful; as already mentioned, in some cases correlations exist between TVP, RVP and temperature.
FLAMMABILITY
General
In the process of burning, hydrocarbon gases react with the oxygen in the air to produce carbon dioxide and water. The reaction gives enough heat to form a flame which travels through the mixture of hydrocarbon gas and air. When the gas above a liquid hydrocarbon is ignited the heat produced is usually enough to evaporate sufficient fresh gas to maintain the flame, and the liquid is said to burn; in fact it is the gas which is burning and is being continuously replenished from the liquid.
Flammable Limits
A mixture of hydrocarbon gas and air cannot be ignited and burn Unless its composition lies within a range of gas in air concentrations known as the 'flammable range'. The lower limit of this range, known as the lower flammable limit (LFQ, is that hydrocarbon concentration below which there is insufficient hydrocarbon gas to support and propagate combustion. The upper limit of the range, known as the upper flammable limit (UFL), is that hydrocarbon concentration above which there is insufficient air to support and propagate combustion.
The flammable limits vary somewhat for different pure hydrocarbon gases and for the gas mixtures derived from different petroleum liquids. Very roughly the gas mixtures from crude oils, motor aviation gasolines and natural-gasoline type products can be represented respectively by the pure hydrocarbon gases propane, butane and pentane. Table 15-1 gives the flammable limits for these three gases. It also shows the amount of dilution with air needed to bring a mixture of 50% by volume of each of these gases in air down to its LFL; this type of information is very relevant to the ease with which vapors disperse to a non-flammable concentration in the atmosphere.
![]()
![]()
 Flammable limits %
vol. Number of dilutions by
Flammable limits %
vol. Number of dilutions by
![]()
![]() Gas hydrocarbon in air air to reduce 50% by
Gas hydrocarbon in air air to reduce 50% by
Upper Lower volume mixture to LFL
![]()
Propane 9.5 2.2 23
Butane 8.5 1.9 26
Pentane 7.8 1.5 33
Table 15-1 Flammable Limits, Propane, Butane, Pentane
In practice the lower-and upper flammable limits of oil cargoes carried in tankers can, for general purposes, be taken as 1 % and 10% by volume respectively.
Effect of Inert Gas on Flammability
When an inert gas, typically flue gas, is added to a hydrocarbon gas/air mixture the result is to increase the lower flammable limit hydrocarbon concentration and to decrease the upper flammable limit concentration. These effects are illustrated in Fig. 15-1, which should be regarded only as a guide to the principles involved.
Every point on the diagram represents a hydrocarbon gas/air/inert gas mixture, specified in terms of its hydrocarbon and oxygen contents. Hydrocarbon gas/air mixtures without inert gas lie on the line AB, the slope of which reflects the reduction in oxygen content as the hydrocarbon content increases. Points to the left of AB represent mixtures with their oxygen content further reduced by the addition of inert gas.
The lower and upper flammability limit mixtures for hydrocarbon g as in air are represented by the points C and D. As the inert gas content increases, the flammable limit mixtures change as indicated by the lines CE and DE, which finally converge at the point E. Only those mixtures represented by points in the shaded area within the loop CED are capable of burning.
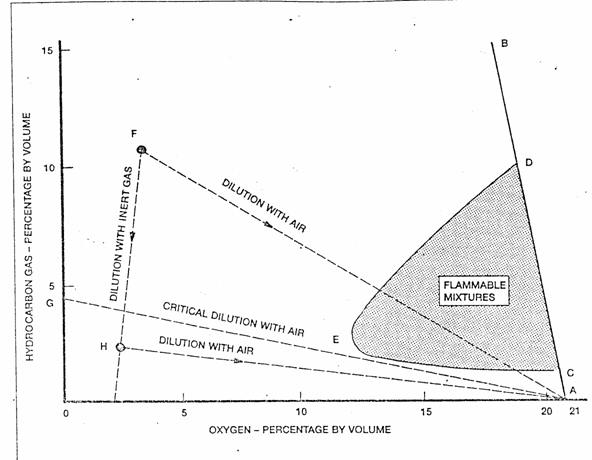
Figure 15-1. Flammability Composition Diagram - Hydrocarbon Gas/Air/Inert Gas
Mixture
This diagram is illustrative only and should not be used for deciding upon acceptable gas compositions in practical cases.
On such a diagram, changes of composition due to the addition of either air or inert gas are represented by movements along straight lines directed either towards the point A (pure air), or towards a point on the oxygen content axis corresponding to the composition of the added inert gas. Such lines are shown for the gas mixture represented by the point F.
It is evident from Fig. 15-1 that as inert gas is added to hydrocarbon gas/air mixtures the flammable range progressively decreases until the oxygen content reaches a level, generally taken to be about 11 % by volume, when no mixture can burn. The figure of 8% by volume of oxygen specified in this guide for a safely inerted gas mixture allows a margin beyond this value.
When an inerted mixture, such as that represented by the point F, is diluted by air its composition moves along the line FA and therefore enters the shaded area of flammable mixtures. This means that all inerted mixtures in the region above the line GA go through a flammable condition as they are mixed with air, for example during a gas freeing operation. Those below the line GA, such as that represented by point H, do not become flammable on dilution. Note that it is possible to move from a mixture such as F to one such as H by dilution with additional inert gas (i.e. purging to remove hydrocarbon gas).
Tests for Flammability
Since hydrocarbon gas/air mixtures are flammable within a comparatively narrow range of concentrations of hydrocarbon gas in air, and concentration, in air is dependent upon vapor pressure, it should in principle be possible to evolve a test for flammability by measuring vapor pressure. In practice, the very wide range of petroleum products and the range of temperatures over which they are handled have prevented the development of one simple test for this purpose.
Instead the oil industry makes use of two standard methods. One is the Reid Vapor Pressure test (see Section 1.2) and the other is the flashpoint test, which measures flammability directly. However, with some residual fuel oils it has been shown that the flashpoint test will not always provide a direct indication of flammability (see Chapter 24).
Flashpoint
In this test a sample of the liquid is gradually heated in a special pot and a small flame is repeatedly and momentarily applied to the surface of the liquid. The flashpoint is the lowest liquid temperature at which the small flame initiates a flash of flame across the surface of the liquid, thereby indicating the presence of a flammable gas/air mixture above the liquid. For all oils, except some residual fuel oils, this gas/air mixture corresponds closely to the lower flammable limit mixture.
There are many different forms of flashpoint apparatus but they fall into two classes. In one the surface of the liquid is permanently open to the atmosphere as the liquid is heated and the result of such a test is known as an 'open cup flashpoint. In the other class, the space above the liquid is kept closed except for brief moments when the initiating flame is introduced through a small port. The result of this class of test is termed a 'closed cup flashpoint.
Because of the greater loss of gas to atmosphere in the open cup test the open cup flashpoint of a petroleum liquid is always a little higher (by about 6 C) than its closed cup flashpoint. The restricted loss of gas in the closed cup apparatus also leads to a much more repeatable result than can be obtained in open cup testing. For this reason, the closed cup method is now more generally favored and is used in this guide in the classification of petroleum. Open cup test figures, however, may still be found in the legislation of various national administrations, in classification society rules and other such documents.
Flammability Classification of Petroleum
There are many schemes for dividing the complete range of petroleum liquids into different flammability classes based on flashpoint and vapor pressure and there is a considerable variation in these schemes between countries. Usually the basic principle is to consider whether or not a flammable equilibrium gas/air mixture can be formed in the space above the liquid when the liquid is at ambient temperature.
Generally in this guide it has been sufficient to group petroleum liquids into two categories entitled non-volatile and volatile, defined in terms of flashpoint as follows:
Non-volatile
Flashpoint of 60 C or above as determined by the closed up method of testing. These liquids produce, when at any normal ambient temperature, equilibrium gas concentrations below the lower flammable limit. They include distillate fuel oils, heavy gas oils and diesel oils. Their RVPs are below 0.007 bar and are not usually measured.
Volatile
Flashpoint below 60 C as determined by the closed cup method of testing. Some petroleum liquids in this category are capable of producing an equilibrium gas/air mixture within the flammable range when in some part of the normal ambient temperature range, while most of the rest give equilibrium gas/air mixtures above the upper flammable limit at all normal ambient temperatures. Examples of the former are jet fuels and kerosenes and of the latter gasolines and most crude oils. In practice, gasolines and crude oils are frequently handled before equilibrium conditions have been attained and gas/air mixtures in the flammable range may then be present.
The choice of 60 C as the flashpoint criterion for the division between non-volatile and volatile liquids is to some extent arbitrary. Since less stringent precautions are appropriate for non-volatile liquids it is essential that under no circumstances is a liquid capable of giving a flammable gas/air mixture ever inadvertently included in the non-volatile category. The dividing line must therefore be chosen to make allowance for such factors as the misjudging of the temperature, inaccuracy in the flashpoint measurement and the possibility of minor contamination by more volatile materials. The closed cup flashpoint figure of 60 C makes ample allowances for these factors and is also compatible with the definitions adopted internationally by the International Maritime Organization (IMO) and by a number of regulatory bodies throughout the world. (See Chapter 24 for information on the relationship between the flashpoint and flammability of residual fuel oils.)
DENSITY OF HYDROCARBON GASES
The densities of the gas mixtures evolved from the normal petroleum liquids, when undiluted with air, are all greater than the density of air. Layering effects are therefore encountered in cargo handling operations and can give rise to hazardous situations.
The following table gives gas densities relative to air for the three pure hydrocarbon gases, propane, butane and pentane, which represent roughly the gas mixtures that are produced respectively by crude oils, by motor or aviation gasolines and by natural gasolines. These figures are not significantly changed if inert gas is substituted for air.
![]()
 Density
relative to air
Density
relative to air

Gas 50% by volume
Pure hydrocarbon hydrocarbon/50% by Lower flammable
volume air limit mixture
![]()
Propane 1.55 1.25 1.0
Butane 2.0 1.5 1.0
Pentane 2.5 1.8 1.0
Table 15-2 Propane, Butane, and Pentane: Densities Relative to Air
It will be seen that the density of the undiluted gas from a product such as motor gasoline is likely to be about twice that of air, and that from a typical crude oil about 1.5 times. These high densities, and the layering effects that result from them, are only significant while the gas remains concentrated. As it is diluted with air the density of the gas/air mixture from all three types of cargo approaches that of air, and at the lower flammable limit is indistinguishable from it.
|
Politica de confidentialitate | Termeni si conditii de utilizare |

Vizualizari: 6213
Importanta: ![]()
Termeni si conditii de utilizare | Contact
© SCRIGROUP 2025 . All rights reserved